| A more fancy copy of this document with dark source code background is available at GIntro README This document describes mainly the use of these bindings for GTK3. For GTK4 you may also consult the pre-print of the Nim GTK4 book located at http://ssalewski.de/gtkprogramming.html. |
| Please do not use the code from the examples in this page, but use the actual code from https://github.com/StefanSalewski/gintro/tree/master/examples . The github readme.adoc does not allow to insert code from files, so I had to manually insert it, and so that code in the html page may not compile with latest gintro version. I plan to create a new page hosted somewhere else with code inserted from files directly. |
This work is partly based on earlier works of J. Mansour and has been supported by E. Bassi and other GTK/Gnome developers.
The combinatorics module was kindly provided by R. Behrends.
|
| This is finally version 0.9.9 of the gintro Nim GTK bindings. It contains some small fixes, that we applied in the last months. Note that gintro’s version numbers are unsigned integers and so wrap around, so whenever there should be one more new release, that may get the tag 0.1 again. (More seriously, we hesitate to call next gintro version 1.0 already, because 1.0 would indicate some final state, which gintro can never archive. All the GTK related libraries are so complex, that it is nearly impossible to create perfect bindings.) But as the number of serious gintro users is tiny, we may fully remove gintro from GitHub by end of 2022. This will save us some work of continous bug fixes, and users can choose one of the 20 other Nim GUI toolkits. It is indeed very questionable if gintro would work with Nim 2.0 or GTK 5.0 at all. And after having worked on gintro for more than 1600 hours, it may be a good decision to just retire now. |
| Version 0.9.8 of the gintro Nim GTK bindings contains some small fixes, including a fix for the latest uref/unref name change in gobject. |
| In version 0.9.7 of the gintro Nim GTK bindings we tried to fix an issue resulting from use of symbols without a module prefix in the code generated by the mconnect() macro. See https://github.com/StefanSalewski/gintro/issues/188. Unfortunately this fix may break some existing projects. We tried hard to make changes as tiny as possible: We tried to let the content of the generated modules unchanged, and changed only files gimplgob.nim and gen.nim and the generated gisup4.nim and gisup3.nim files. The example programs still compile and seems to run. |
| In version 0.9.6 of the gintro Nim GTK bindings we tried to fix a bug related to the appearance of libsoup 3.0. When gobject-introspection was first processing libnice, it loads old libsoup 2.4 and when then processing linsoup 3.0 name conflicts lead to error messages and the install process hung. Now we process always libsoup in version 2.4 and 3.0 before libnice, this seems to work. For libsoup 2.4 we generate a module called libsoup.nim as before, and the libsoup 3.0 module is now called libsoup3.nim. |
| For version 0.9.5 of the gintro Nim GTK bindings we added a patch to support very old GTK3 versions like Debian Buster, updated the list of "light symbols" like Rectangle, TextIter and such that do not need Nim Proxy objects, and finally fixed a recent glib proc name conflict. |
| Version 0.9.4 of the gintro Nim GTK bindings contains a lot of fixes. Before 0.9.4 some GtkDrawingArea examples gave crashes with the most recent Nim compiler due to a wrong cast from ref object to RootRef. That is fixed, and the drawingarea example works now also without manually freeing cairo resources like Context, Surface and Pattern. We have not updated the example, so it still contains the manually memory management, but you can comment it out if you want. When you compile your code with --gc:arc you really should not have to care for releasing cairo resources. If you still use the default refc GC you may release cairo resources manually, as the GC has a delay, which may first allocate some GB before the GC becomes active and frees the cairo resources. Note that only a small part of all the cairo functions has been tested yet, so there may still be bugs. Strings are now passed to GTK functions as cstrings, so you can pass nil as well as empty strings. The nil value has for GTK in most cases a special meaning and is different from an empty "" string. For some function parameters nil is the default value for cstrings. Finally we support now named empty flag sets, so instead of passing the empty set as {} for default flag set values you can use names like BindingFlagsDefault or BindingFlags.default. |
| Version 0.9.3 of the gintro Nim GTK bindings contains some serious changes following the discussion in https://discourse.gnome.org/t/get-ref-function-for-none-gobject-classes-like-gtkexpression/6696. With that changes issue https://github.com/StefanSalewski/gintro/issues/135 should be fixed, and the listview_clocks example works with a C and a Nim part. For most apps that changes should be invisible, but it is possible that we have introduced new bugs or maybe memory leaks. At least the existing examples seems still to compile, but we have not yet tested them all. And the notify signals for gobject properties like "notify::cursor-moved" for entry widgets should work now. |
| The version 0.9.2 of the gintro Nim GTK bindings is only a fix for the issue with latest gstreamer from gitlab, see https://github.com/StefanSalewski/gintro/issues/138. |
| Version 0.9.1 is mostly a plain fix for issue https://github.com/StefanSalewski/gintro/issues/133. For GObject proc parameters with direction in and transfer full we have to avoid that the Nim memory management destroys the GObject when the Nim proxy object is destroyed. Current fix was adding a plain ".ignoreFinalizer = true" for that case, which works in most cases. But maybe a better fix would be to ref() the gtk object instead. Maybe that makes no difference for gobjects, but it can make a difference for entities like GtkExpression which are used further, see issue https://github.com/StefanSalewski/gintro/issues/137. But we will leave that for next version 0.9.3. |
| Due to a user request we added support for the adwaita library for version 0.8.9. This lib is the libhandy variant for GTK4 and is intended to support GTK on mobile devices. Unfortunately we can not yet provide an example program for this library. The C example from git sources is not really tiny, so porting to Nim would take some hours at least, as we have absolutely no knowledge about that lib yet. Maybe we can provide an example next year or maybe that user will finally provide something? |
| Starting with version 0.8.8 of the gintro Nim GTK bindings for procs like getStartIter() without a result but with a var out parameter an overloaded version is created where the var out parameter is returned as result. So we can write "let startIter = buffer.getStartIter()" now. And we tried to fix the issues with out gobject parameters as in g_file_new_tmp(). |
| For version 0.8.7 of the gintro Nim GTK bindings we did a larger internal cleanup for the gen.nim generator script but tried to generate output modules identical to v.0.8.6 still. For next release v0.8.8 there will be some changes in the generated modules then. Also for this version 0.8.7 we do support webkit2 for GTK4. |
| Version 0.8.6 of the gintro Nim GTK bindings contains now all the standard gst modules, and due to a recent request also the gtklayershell.nim module. Due to issues with some gst modules we do now call init() for the gst module before we use it with gobject-introspection. We do the same for GTK3 and GTK4 as these provide also an init() proc. According to a recent discussion with the GTK core devs that init() call is necessary. The call is done by use of dlopen(), for which we need to provide the names of the dynamic libraries, which is some guesswork for Windows and Mac. |
| For version 0.8.5 of the gintro Nim GTK bindings we have added webkitgtk support. Unfortunately still only for GTK3, as the latest webkitgtk package 2.30.4 does not compile with stable GTK4. But we should get webkitgtk for GTK4 in a few weeks. One question is still how to name the GTK4 version then. The official name of the version for GTK3 is webkit2, so shall we call the version for GTK4 webkitgtk4 or webkit4? Or different? |
| Since version 0.8.4 we do support the libnice module for Linux and Windows. And we added gtksourceview5 to support gtksourceview usage for GTK4. The module is called gtksourceview5 because the underlying C library has mayor version 5 already. So you can write your own GTK4 Nim editor now. Additional optional var out parameters are supported now and GtkGestures should work also. |
| The version gintro v0.8.3 will have some internal cleanup, which removes the temporary Array types. In the past we used that names to indicate array parameters, it was working well, but still it was ugly using names to indicates data types. So we fixed that. And we have added some better support for GList parameter. GLists are now converted to Nim seqs and vice versa. Not everywhere still, and this conversion is still untested, as GList parameters are used most of the time in exotic functions only. Another change is that we have support for libnice by a request of a user from Japan now. As he intents to use libnice on Windows 8.1 with only glib and gobject installed, but with no gtk installed, we had to break module gimpl.nim into two units called gimplgobj.nim and gimplgtk.nim, and we added a module called dummygtk.nim which can be imported to make the connect() macro available while a real gtk module is not available. Please let us know if these changes should break something for ordinary gtk users. To test libnice we added the sdp_example.nim converted from a C example, it seems to compile and run. But we have not tested real communication between different computers yet, and not tested it on windows at all. If you have a use case for libnice you may test it yourself, possible issues may Nim’s string vs seq[uint8] vs plain ptr char, or the uint data types in C example which we replaced by plain int as used in Nim prefereable. Debugging should be not hard, if you do not manage it yourself then you may open a github issue. Version v0.8.3 uses also some more light entities like gtk.TextIter or glib.Mutex, that are entities which are generally allocated on the stack and are only initialized to plain binary zero. For these types no proxy symbols are needed. Unfortunately discovering these entities by use of gobject-introspection works not reliable, so we are using a manually created list, which may contain errors. And finally with v0.8.3 the examples from the GTK4 book (http://ssalewski.de/gtkprogramming.html) should work. For the GTK3 examples we had to do some minimal fixes, so you may have to do similar fixes for your own code as well. |
| Version 0.8.2 of gintro was only a fix for recent gstreamer 1.18 which added some uncommon gobject-introspection stuff which brook the install of 0.8.1 for a few users. We were not yet able to verify that the gst example which is bases on the gst C tutorial1 works with gstreamer 1.18. |
| For version 0.8.1 of gintro, requested by FedericoCeratto, we have added libhandy support. Libhandy is not yet available for GTK4 but seems to work with gtk+-3.24.22. As Gentoo Linux ships still only a very old libhandy, we tested with latest release from https://gitlab.gnome.org/GNOME/libhandy/ Only available example is currently /examples/gtk3/handy.nim converted from example.py. |
| If you are using Arch Linux you may still have to ensure that curl or wget is available, see https://github.com/StefanSalewski/gintro/issues/83. Next release will get support for GList proc parameters and results, but providing that is some non trivial work and may break some other stuff unfortunately. Note that the File type of module gio is now called GFile to prevent name conflicts with Nims own File type. |
| Version 0.7.7 contains the fixes from https://github.com/StefanSalewski/gintro/issues/75. Mr lscrd has tested it already by use of nimble install gintro@#head some weeks ago, so we may assume that it works properly. Next version will again have serious modifications for GTK4, see http://ssalewski.de/gtkprogramming.html. So it may be a good idea to have a version 0.7.7 available as a fallback. |
| Starting with version 0.7.4 we support latest GTK 3.98.3 which may become GTK 4 at the end of 2020. |
Starting with version v0.7.3 we allow passing a type descriptor as first parameter to the
new() procs like newButton(MyButtonSubclass) to support subclassing and extending Widgets in OOP style.
See Extending or sub-classing Widgets for an complete example. The init() procs which were used
before for this task are now deprecated and will be removed later. This new approach generally saves
one line of source code, allows using let instead of var, and the naming of procs is more consistent.
|
Starting with version v0.7.1 we have added destructor support when compiled with --gc:arc,
so we have no memory leak for subclassed objects any more, see the example in Extending or sub-classing Widgets section.
When compiled with default (refc) GC finalizers are used as before. And for objects marked with nullable
tag in gobject-introspection we now return nil value for the proxy object when the C lib has returned NULL.
So according to the C API docs, you can check for nil result for the few functions which may return NULL.
Nil objects returned by GTK Builder causes now a program termination (by assert()) because in that case
nil should indicate a programming error.
|
Starting with version v0.7.0 we support the new Nim memory management called ARC, see
https://forum.nim-lang.org/t/5734. Just compile your programs with --gc:arc. The main advantage is that
ARC is deterministic, so it is easier to find bugs in the bindings or in your programs. And
manually freeing resources, as we did previous for some cairo data structures to free them
without GC delay should be now unnecessary. Generally this version should be more stable:
Nim without option --gc:arc compiled new() calls silently with and without a finalizer proc parameter for
the same data type, but the finalizer was then always called. This behaviour was stated in the Nim manual,
but it was easy to forget this strange behaviour, so unintentionally finalizer calls may have ocurred.
Nim with --gc:arc detects at least some of these errors, and for gintro we now try hard to not mix these calls.
Generally we specify a finalizer, and use a field in the ref object to ignore the call when necessary.
For a type always the same finalizer has to be used (or always none) and finalizer must be defined in the
same module as the object type itself. For this to work reliable we have generally to qualify the
finalizer proc with its module prefix. All that made a larger rewrite of gen.nim generator script
necessary, with the danger of introducing bugs. We have not tested v0.7 much yet, the examples in
gtk3 directory compile and seems to start at least. We would still have to check the macros in gimpl.nim more
carefully — we had to replace deepcopy by a plain copy and removed a (wrong?) GC_ref(). Generally we have
to investigate possible memory leaks. One leak is unavoidable: If we subclass Widgets, then
finalizer are not applied to the GTK object, so its memory leaks. See https://forum.nim-lang.org/t/5825#36241.
But that should be not a too serious problem, subclassed objects are generally only allocated once
in a program and generally live as long as the program is running any way. For the next version of
gintro we do consider using only destructors and no finalizers, see https://forum.nim-lang.org/t/5854
and https://forum.nim-lang.org/t/5786. That may simplify the code and enable subclassed GTK objects
to release its memory, but require rewriting gen.nim again. But then we would have to use --gc:arc
always. Maybe we can join both by specifying some conditional when expression — we will see.
If for you installation or compiling with v0.7 should not work, then please report issues on
github issue tracker and continue using v0.6.1 for now.
|
Starting with version v.0.6.0 we support gstreamer (gst). At the same time we have split
cairo module into an gobject-introspection basic part and an manually created part. Unfortunately the
gobject-introspection is not available for very old GTK/cairo libraries, so installation may fail for you.
Use v0.5.5 in this case. Also we support gBoxed types now, this is assumed to work well but is not well tested yet.
Command to install older releases should be something like
nimble uninstall gintro followed by nimble install gintro@v0.5.5
|
| Starting with release 0.5.3 we do not generate field entries for objects and we do not generate class structs and private objects. Also we stopped exporting the low level functions like gtk_button_new(). For a real high-level binding we should not need these. If that is a serious limitation for you, then use release 0.5.2 for now and create an github issue for your use case, we will try to fix it, maybe undo these changes. Also starting with v0.5.3 we try to support array parameters like TargetEntryArray, PageRangeArray and KeymapKeyArray. Use of these array parameters is rare, if you will use functions with these parameters you may inspect the source code first, as the code is auto-generated and still untested. |
| Starting with release 0.5.0 we also support GTK4. GTK4 is still work in progress and not intended for end users yet, but it is good to have it available for migration testing. For GTK4 we have a new module gsk, and new versions of modules gtk, gdk and gdkX11, which are not backward compatible with the old once of GTK3. The other modules can be used by GTK3 and GTK4 in parallel. Due to this fact we use a single nimble package which can be used for GTK3 and GTK4 development. To archive this, we have named the new modules gtk4, gdk4 and gdkX114 — the old once are named gtk, gdk and gdkX11 still. So for existing GTK3 software no code changes are necessary. For GTK4 an example is provided — it imports gtk4 instead of gtk now, and instead of window.showAll() window.show() is needed. More GTK4 examples may follow eventually, see GTK4 migration page at https://developer.gnome.org/gtk4/stable/gtk-migrating-3-to-4.html. The gintro package tries to install the GTK4 modules when GTK4 is available on the local computer and skips it if not available. For successful detection of GTK4 the typelibs must be found. For example, if you have installed GTK4 from sources on /opt/gtk as described in https://developer.gnome.org/gtk4/3.96/gtk-building.html, then you may have to execute "export GI_TYPELIB_PATH=/opt/gtk/lib64/girepository-1.0" in your shell before you do "nimble install gintro". Currently gtksourceview and vte is not available for GTK4. GTK4 provides a official test program called gtk4-demo — of course that one should work fine on your box before you consider testing Nim with GTK4. |
Nim is a modern universal programming language.
GTK, also known as the Gimp Tool Kit and now sometimes called Gnome Tool Kit, is a Graphical User Interface library.
| Later we will insert at this location a nice picture of a fancy Nim GTK3 GUI. Such a picture is fine to attract users and indeed is a good motivation. But such pictures are no real evidence for the quality of a GUI toolkit — the concrete example may look nice, while the toolkit looks much worse in other environments and offers by far not all that what is needed in real life. |
While GTK was initially designed and advertised as cross platform GUI toolkit, it is currently mostly used on Linux and other Unix like operation systems. Most Linux distributions include it, and some use it for their default desktop environment, often with the Gnome environment or other window managers. While GTK2 applications like GIMP are still used on Windows, there seems to exist currently only very few GTK3 applications for Windows or MacOSX. When you develop primary free open source software (FOSS) for Linux or other Unix like operating systems, then GTK3 is a good choice for you. With some effort you should be even able to port your application to the proprietary Windows or MacOSX operating systems. But when your primary target platforms are Windows and MacOSX and you desire a real native look and feel there, then you may find better suited ones in the Nim software repository. Also, when you only need a minimal restricted GUI which is very easy to install on Windows and MacOSX, then you may find better suited packages in the Nim package repository. Android OS is currently not supported by GTK at all.
| At least for Windows 10 it seems to be not that hard to install GTK3 libraries, as was recently reported in https://github.com/StefanSalewski/gintro/issues/24 by user zetashift: |
Sketch of GTK3 install for Windows 10: For the GTK libs I did according these instructions(https://www.gtk.org/download/windows.php): Install MSYS2 In the msys2 cmd I entered: pacman -S mingw-w64-x86_64-gtk3 Then for some other necessary depencies(girepository.dll) you need to do: pacman -S mingw-w64-x86_64-python3-gobject Additional, you have to install the separate GtkSourceView lib in a similar manner from https://github.com/Alexpux/MINGW-packages/blob/master/mingw-w64-gtksourceview3/
While low level Nim bindings for GTK3 are already available since a few years, this one is an attempt to provide real high level bindings with full type safety, full Garbage Collector (GC) support and an idiomatic Application Programming Interface (API).
Currently there are at least 3 sources of GTK3 bindings for Nim:
-
https://github.com/ngtk3 (obsolete, has been deleted)
ngtk3 was the first attempt to provide GTK3 support for Nim. It contained single repositories for all the GTK related libraries and was not supported by nimble package manager. It was created from GTK 3.20 headers and is now deprecated.
oldgtk3 is the port of ngtk3 to GTK 3.22 — joining all libraries and providing nimble support. Some people may still prefer using oldgtk3. As it is generated with the Nim tool c2nim directly from the C header files without much manual intervention, it should be complete and contain not that much bugs. Missing Garbage Collector support is generally not really a problem, as widgets are generally put into containers and were automatically deleted together with its parents due to GTK’s reference counting.
Still there can be some demand for really high level bindings — so this gintro repository tries to provide them.
High level GTK3 bindings, as available for many other programming languages like C++, Python, Ruby or D already, have these advantages:
-
full Garbage Collector or Destructor support — you should never have to free resources manually
-
Widgets are Nim objects, so inheritance and sub-classing can be used
-
full type safety — no needs for casts or other unsafe and dangerous operations
These high level bindings are based on GObject-Introspection, an XML based database like interface description. Compared to the C header files this description gives us more and deeper information about data types and function calls, for example ownership transfer of objects and in or out direction of procedure variables, which makes writing the glue code much easier. And it should work with minimal modifications also for the upcoming GTK4.
Unfortunately there are also some drawbacks:
-
The Application Programming Interface (API) will be different from what is known from C API, so using C examples or C tutorials is not really straight forward
-
The high level source code will differ from available C examples, so there would be a big demand for tutorials
-
We need a lot of glue code, which has much room for bugs. So much testing is necessary.
-
There is some overhead due to indirect calls, leading to some code size increase and minimal performance loss.
| The new package name is gintro, short for GObject-Introspection. The previous name was nim-gi, but the hyphen is deprecated for package names, as is the nim prefix. |
Current state of these bindings
We are still in an early stage, but it is already more than a proof of concept. GTK and related libraries have many thousand of
callable functions and nearly as many data types. Testing all that is nearly impossible for a small team with limited resources.
The initial approach was to generate low level
bindings, which looked similar to the ones generated by the c2nim tool from the C headers. After that was done, we have associated all
the C structs and GObject data types with Nim proxy objects. A well defined relation between these proxy object and the low level C data types
should ensure fully automatic garbage collection. This is supported by smart type conversion, for example C strings returned by glib library
are assigned to newly created Nim strings, while the memory of the C strings is automatically freed. For most cases this seems to work. But there
exists a few more complicated cases, for example functions may return whole arrays of C strings or other non elementary data types,
or function arguments or results may be so called glists,
list structures of glib library. These cases can not be processed automatically but needs carefully manual investigations. And there may be still functions and data
types missing: GObject-Introspection query gives us many thousand lines of Nim interface code, and it is not really obvious if and what is missing.
Some functions and data types are missing for sure — at least some low level ones, which are considered unneeded for high level bindings by GObject-Introspection.
But maybe more is missing, we have to investigate that. Until now these bindings have been tested only for 64 bit Linux systems with GTK 3.24.
These basic libraries are already partly tested:
Gtk, Gdk, GLib, GObject, Gio, GdkPixbuf, GtkSource, Pango, PangoCairo, PangoFT2, GModule, Rsvg, fontconfig, freetype2, xlib, Atk, Vte, cairo
In best case it should be possible to add more GObject based libraries to this list without larger modifications of the generator source code. Unfortunately the bindings for the cairo drawing library provided by GObject-Introspection was only a minimal stub — we have extend it manually.
How to try it out
Of course you will need a working Nim installation with a recent compiler version and you have to ensure that GTK and related libraries are installed on your system. For some Linux distributions which provide mainly pre-compiled software you may have to also install some GTK related developer files.
With a recent nimble version (>= v0.8.10) you only have to type in a shell window:
nimble install gintro
| Latest version of gintro package uses some files from oldgtk3 package for bootstrapping. We assume that users of gintro generally are not interested in low level oldgtk3 package, so we try to download only 3 single files from oldgtk3 package. That should work if wget or nimgrab executables are available. If it fails you should get a longer error message which may help you to solve the issue. |
Nimble prepare should run for about 20 seconds, it compiles and executes the generator program gen.nim.
Unfortunately we can not guarantee that the generator command will be able to really build all the
desired modules. The built process highly depends on your OS and installed GTK version. For 64 bit Linux systems
with GTK 3.24 and all required dependencies installed it should work. For never GTK versions it may fail, when that GTK
release introduces for example new unknown data types like array containers. In that case manual fixes may be necessary.
The GObject-Introspection based built process generates bindings customized to the OS where the generator is executed,
so for older GTK releases or a 32 bit system different files are created. Later we may also provide pre-generated
files for various OS and GTK versions, but building locally is preferred when possible.
|
A few basic examples
| Currently we do not install the example programs. If you want to try them, you have to copy the source code of the examples from https://github.com/StefanSalewski/gintro/tree/master/examples to your local computer, maybe to /tmp/gintro/examples directory. |
Then you can compile and run them from shell with commands like
cd /tmp/gintro/examples/ nim c app0.nim ./app0
or you may open the source files in your favorite Nim IDE or editor. Taking the source code from this Readme file is not really recommended, as these source code listings may be not the latest versions.
GTK3 programs can use still the old GTK2 design, where you first initialize the GTK library, create your widgets and finally enter the GTK main loop. This style is still used in many tutorials as in Zetcode tutorial or in the GTK book of A. Krause. Or you can use the new GTK3 App style, this is generally recommended by newer original GTK documentation. Unfortunately the GTK3 original documentation is mostly restricted to the GTK3 API documentation, which is generally very good, but makes it not really easy for beginners to start with GTK. API docs and some basic introduction is available here:
If you should decide to continue developing software with GTK, then you may consider installing the so called
devhelp tool. It gives you easy and fast access to the GTK API docs. For example, if you want to use a Button Widget in your
GUI and wants to learn more about related functions and signals, you just enter Button in that tool and are guided to
all the relevant information.
|
We start with a minimal traditional old style example, which should be familiar to most of us:
# nim c t0.nim
import gintro/[gtk, gobject]
proc bye(w: Window) =
mainQuit()
echo "Bye..."
proc main =
gtk.init()
let window = newWindow()
window.title = "First Test"
window.connect("destroy", bye)
window.showAll
gtk.main()
main()This is the traditional layout of GTK2 programs. When using this style then it is important to initialize the GTK library by calling gtk.init()
at the very beginning. Then we create the desired widgets, connect signals, show all widgets and finally enter the GTK main loop
by calling gtk.main. About connecting signals we will learn more soon, for now it is only important that we have to connect to
the destroy signal here to enable the user to terminate program execution by clicking the window close button.
Now a really minimal but complete App style example, which displays an empty window.
| The source text of all these examples is contained in the examples directory. Unfortunately github seems to not allow to include that sources directly into this document, so there may be minimal differences between the source code displayed here and the sources in examples directory. |
# app0.nim -- minimal application style example
# nim c app0.nim
import gintro/[gtk, glib, gobject, gio]
proc appActivate(app: Application) =
let window = newApplicationWindow(app)
window.title = "GTK3 & Nim"
window.defaultSize = (200, 200)
showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()In the main proc we create a new application and connect the activate signal to our activate proc, which then creates and displays
the still empty window.
We are importing modules gtk and gio. Initially both modules had a data type called Application (gtk.Application
extends indeed the gio.Application), so we would have to use module name prefixes, or we could import from gio only
what is really needed (from gio import …) or use the form (import gio exept …). But as gio.Application is generally
not needed often, we have no renamed gio.Application to GApplication. No more name clashes.
|
Various ways to set widget parameters are supported — the number 1 to 6 refer to the comments below:
setDefaultSize(window, 200, 200) (1)
gtk.setDefaultSize(window, 200, 200) (2)
window.setDefaultSize(200, 200) (3)
window.setDefaultSize(width = 200, height = 200) (4)
window.defaultSize = (200, 200) (5)
window.defaultSize = (width: 200, height: 200) (6)| 1 | proc call syntax |
| 2 | optional qualified with module name prefix |
| 3 | method call syntax |
| 4 | named parameters |
| 5 | tupel assignment |
| 6 | tupel assignment with named members |
Well, that empty window is really not very interesting. The GTK and Gnome team provides some GTK examples at https://developer.gnome.org/gnome-devel-demos/. The C demos seems to be most actual and complete, and are easy to port to Nim. So we start with these, but if you are familiar with the other listed languages, then you can try to port them to Nim as well. Let us start with https://developer.gnome.org/gnome-devel-demos/3.22/button.c.html.en as it is still short and easy to understand, but shows already some interesting topics.
The C code looks like this:
Converting it to Nim is straight forward with some basic C and Nim knowledge, and Nim does not force us
to convert its shape into all the classes known from pure Object Orientated (OO) languages. We can either use the
Nim tool c2nim to help us with the conversion, or do it manually. Indeed c2nim can be very helpful by
converting C sources to Nim. Most of the time it works well. Personally I generally pre-process C files, for example
by removing too strange macros and defines, or by replacing strange constructs, like C `for loops, to simpler
ones like while loops. Then I apply c2nim to the C file and finally manually compare the result line by line and
fine tune the Nim code. But for this short source text we may do all that manually and finally get something like
this:
Again we have the basic shape already known from app0.nim example: Main proc creates the application, connect
to the activate signal and finally runs the application. When GTK launches the application and emits the activate signal, then
our activate proc is called, which creates a main window containing a button widget. That button is again connected with a
signal, in this case named clicked. That signal is emitted by GTK whenever that button is clicked with the mouse and results
in a call of our provided buttonClicked() proc. The procs connected to signals are called callbacks and generally got the widget
on which the signal was emitted as first parameter. They can also get a second optional parameter of arbitrary type — we will
see that in a later example. This callback here gets only the button itself as parameter, and it’s task is to reverse the
text displayed by the button. Not very interesting basically, but we are indeed using the glib function utf8Strreverse()
for this task. While that function internally works with cstrings, and in C we have to free the memory of the returned cstring,
in our Nim example that is done automatically by Nim’s Garbage Collector. When you compare our example carefully with the C code,
then you may notice a difference. The C code passes the window containing the button as an additional parameter to the
callback function, but that parameter is not really used. We simple ignore it here, as it is not used at all.
In one of the following examples you will learn how passing (nearly) arbitrary parameters in a type safe way is done.
Another difference is, that the C code returns an integer status value returned by g_application_run() to the OS. We
could do the same by using the quit() proc of Nim’s OS module, but as that would give us no additional benefit, we simply ignore it.
The command nim c sourcetext.nim generates an executable which contains code for runtime checks and debugging,
which increases executable size and decreases performance.
After you have tested your software carefully, you may give the additional parameter -d:release to avoid this. For the gcc backend
you may additional enable Link Time Optimization (LTO), which reduces executable size further. To enable LTO you may put
a nim.cfg file in your sources directory with content like
|
path:"$projectdir" nimcache:"/tmp/$projectdir" gcc.options.speed = "-march=native -O3 -flto -fstrict-aliasing"
With that optimization, your executable sizes should be in the range of about 50 kB only!
Optional, type safe parameters for callbacks
The next example shows, how we can pass (nearly) arbitrary parameters to our connect procs.
We pass a string, an object from the stack, a reference to an object allocated on the heap
and finally a widget (in this case the application window itself, you may also try passing
another button). As the main window itself is a so called GTK bin and can contain only one
single child widget, we create a container widget, a vertical box in this case, fill that box with
some buttons, and add that box to the window.
Compile and start this example from the command line and watch what happens when you click on the buttons.
# nim c connect_args.nim
import gintro/[gtk, glib, gobject, gio]
type
O = object
i: int
proc b1Callback(button: Button; str: string) =
echo str
proc b2Callback(button: Button; o: O) =
echo "Value of field i in object o = ", o.i
proc b3Callback(button: Button; r: ref O) =
echo "Value of field i in ref to object O = ", r.i
proc b4Callback(button: Button; w: ApplicationWindow) =
if w.title == "Nim with GTK3":
w.title = "GTK3 with Nim"
else:
w.title = "Nim with GTK3"
proc appActivate (app: Application) =
var o: O
var r: ref O
new r
o.i = 1234567
r.i = 7654321
let window = newApplicationWindow(app)
let box = newBox(Orientation.vertical, 0)
window.title = "Parameters for callbacks"
let b1 = newButton("Nim with GTK3")
let b2 = newButton("Passing an object from stack")
let b3 = newButton("Passing an object from heap")
let b4 = newButton("Passing a Widget")
b1.connect("clicked", b1Callback, "is much fun.")
b2.connect("clicked", b2Callback, o)
b3.connect("clicked", b3Callback, r)
b4.connect("clicked", b4Callback, window)
box.add(b1)
box.add(b2)
box.add(b3)
box.add(b4)
window.add(box)
window.showAll
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard app.run
main()To prove type safety, we may modify one of the callback procs and watch the compiler output:
proc b1Callback(button: Button; str: int) =
discard # echo strconnect_args.nim(37, 5) template/generic instantiation from here gtk.nim(-15021, 10) Error: type mismatch: got (ref Button:ObjectType, string) but expected one of: proc b1Callback(button: Button; str: int)
It may be not always really obvious what the compiler wants to tell us, but at least we are told that it got a string and expected an int.
Currently the connect function is realized by a Nim type safe macro. Connect accepts two or three
arguments — the widget, the signal name and the optional argument. When the optional argument
is a ref (reference to objects on the heap) then it is passed as a reference, otherwise a deep copy
of the argument is passed. For the above code this means, that r and the window variables are passed
as references, while the string and the stack object are deep copied. Currently it is not possible
to release the memory of passed arguments again. This should be no real problem, as in most
cases no arguments are passed at all, and when arguments are passed, then they are general
small in size like plain numbers or strings, or maybe references to widgets which could not be freed
at all, as they are part of the GUI. Later we may add more variants of that connect macro.
Navigation can be hard for beginners. You may have basic knowledge of GTK and want
to build a GUI for your application. But how to find what you need. Well, we offer no separate
automatically generated API documentation currently, as that is not really helpful. In most cases
it is easy to just guess Nim symbol names, proc parameters and all that. Using a smart editor
with good nimsuggest support further supports navigation — for example NEd shows us
all the needed proc parameters when we move the cursor on a proc name, or we press Ctrl+W and jump
to the definition of that symbol. For unknown stuff the original C function name is often a good starting point.
Assume you don’t know much about GTK’s buttons, but you know that you want to have a button in
your GUI application. GTK generally offers generator functions containing the string new in their name.
So it is easy to guess that there exists a C function named gtk_button_new. That name is also
contained in the bindings files, in this case in gtk.nim. So we open that file in a text editor and search for
that term. So it is really easy to find first starting points for related procs and data types. Most data types
are located near by their related functions, so you should be able to find all relevant information fast.
Remember the GTK devhelp tool, and use also grep or the nimgrep variant.
|
Extending or sub-classing Widgets
It may occur that we want to attach additional information to GTK widgets
by extending or subclassing them. Doing this is supported
by providing for each widget class not only a corresponding new() proc which returns
the newly created widget, but also
a init() proc, which gets an uninitialized variable of the (extended) widget type as argument and
initializes that variable with a newly created
GTK widget.
Doing this is supported
by providing for each widget class an additional new() proc which takes an type descriptor
as first argument, like newButton(CountButton, "Counting down from 100 by 5") in the example below.
Initializing the added fields is
done separately by the user. The following code shows a GTK button, which is
extended with a counter member field. That counter is decreased for
each button click. The amount of decrease (5) is passed to the callback as a int parameter.
Recent tests proved that providing custom destructors is not really needed any more, see https://forum.nim-lang.org/t/7360#46632.
Since gintro version 0.7.1 we support destructors when compile option --gc:arc is used.
To destroy subclassed widgets we have to create a =destroy() proc as shown in the code below.
This may look a bit verbose, and it is only necessary to avoid memory leaks for widgets
which are created and destroyed multiple times during program execution. Most widgets are
created at startup and live until program terminates, so there is no noticeable leak even
without a matching destroy. (In examples/gtk3 there is a extended file called subclassArcDestructorTest.nim
to test the destructor behaviour.)
In this example we have to define our new widget type first, then we have to declare a variable of that type and pass that variable to the init() proc.
CSS styles, GErrors and Exceptions

Often GTK beginners ask how one can apply custom styles to GTK widgets, for example custom colors. While in most cases the use of custom colors gives just ugly results, as the custom colors generally do not match well with the default color scheme, it is good to know how we can do it. For GTK3 styles are applied to widgets by using Cascading Style Sheets (CSS). You may find C example code similar to this:
// https://stackoverflow.com/questions/30791670/how-to-style-a-gtklabel-with-css
// gcc `pkg-config gtk+-3.0 --cflags` test.c -o test `pkg-config --libs gtk+-3.0`
#include <gtk/gtk.h>
int main(int argc, char *argv[]) {
gtk_init(&argc, &argv);
GtkWidget *window = gtk_window_new(GTK_WINDOW_TOPLEVEL);
GtkWidget *label = gtk_label_new("Label");
GtkCssProvider *cssProvider = gtk_css_provider_new();
char *data = "label {color: green;}";
gtk_css_provider_load_from_data(cssProvider, data, -1, NULL);
gtk_style_context_add_provider(gtk_widget_get_style_context(window),
GTK_STYLE_PROVIDER(cssProvider),
GTK_STYLE_PROVIDER_PRIORITY_USER);
g_signal_connect(window, "destroy", G_CALLBACK(gtk_main_quit), NULL);
gtk_container_add(GTK_CONTAINER(window), label);
gtk_widget_show_all(window);
gtk_main();
}Converting that to Nim is again straight forward:
# nim c label.nim
import gintro/[gtk, glib, gobject, gio]
proc appActivate(app: Application) =
let window = newApplicationWindow(app)
let label = newLabel("Yellow text on green background")
let cssProvider = newCssProvider()
let data = "label {color: yellow; background: green;}"
#discard cssProvider.loadFromPath("doesnotexist")
discard cssProvider.loadFromData(data)
let styleContext = label.getStyleContext
assert styleContext != nil
addProvider(styleContext, cssProvider, STYLE_PROVIDER_PRIORITY_USER)
window.add(label)
showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()For this example we create a plain label widget with some text. To colorize it, we generate a CssProvider and load it with a textual description of our desired colors. Then we extract the style context from the label and add our CssProvider to it.
The last parameter of the C function gtk_css_provider_load_from_data() is of type GError and can
be used in C code to detect runtime errors. The C code above just passes NULL to ignore this error.
For Nim we map that GError argument to exceptions. To test what happens in Nim when an GError would
report an error condition, you may uncomment function loadFromPath() in the code above. As the specified path
does not exist, we should get an exception with a message telling us the problem. Of course in your real
code you may catch such exceptions with Nim’s try: blocks. (You may also modify the data variable above to
an illegal CSS statement — if the statement is seriously wrong, then you should get an exception from
loadFromData().
SpinButton
This widget is used for entering numerical values. We can type in the value with the keyboard, click on the +/- symbols or use the scroll wheel of the mouse. This example also shows that we can use vertical or horizontal orientation for this widget, and how we can use bindProperty() to bind a property of one widget to another widget. Here we use a button to control wrapping behaviour of the spin buttons.
GTK Builder — user interfaces created with the glade tool
As C code can be very verbose, some people prefer outsourcing the GUI layout in XML files which can be created and modified with the glade GUI creator program. For high level languages like Python or Nim the program source code is generally short and clean, so that use of XML files may not have much benefit. But of course we can use GTK builder from Nim. We follow the example from https://developer.gnome.org/gtk3/stable/ch01s03.html but we modify it to use the new GTK3 app style: For the XML file we have to change only class="GtkWindow" into class="GtkApplicationWindow". Our Nim program has the well known application shape, with one addition: We have to explicitly set the application for the main window. Of course you can also use the traditional program structure with Nim and Builder, for that case you can straight follow the linked page or other examples. Here is the XML file and the Nim code:
<interface>
<object id="window" class="GtkApplicationWindow">
<property name="visible">True</property>
<property name="title">Grid</property>
<property name="border-width">10</property>
<child>
<object id="grid" class="GtkGrid">
<property name="visible">True</property>
<child>
<object id="button1" class="GtkButton">
<property name="visible">True</property>
<property name="label">Button 1</property>
</object>
<packing>
<property name="left-attach">0</property>
<property name="top-attach">0</property>
</packing>
</child>
<child>
<object id="button2" class="GtkButton">
<property name="visible">True</property>
<property name="label">Button 2</property>
</object>
<packing>
<property name="left-attach">1</property>
<property name="top-attach">0</property>
</packing>
</child>
<child>
<object id="quit" class="GtkButton">
<property name="visible">True</property>
<property name="label">Quit</property>
</object>
<packing>
<property name="left-attach">0</property>
<property name="top-attach">1</property>
<property name="width">2</property>
</packing>
</child>
</object>
<packing>
</packing>
</child>
</object>
</interface> https://developer.gnome.org/gtk3/stable/ch01s03.html
# builder.nim -- application style example using builder/glade xml file for user interface
# nim c builder.nim
import gintro/[gtk, glib, gobject, gio]
proc hello(b: Button; msg: string) =
echo "Hello", msg
proc quitApp(b: Button; app: Application) =
echo "Bye"
quit(app)
proc appActivate(app: Application) =
let builder = newBuilder()
discard builder.addFromFile("builder.ui")
let window = builder.getApplicationWindow("window")
window.setApplication(app)
var button = builder.getButton("button1")
button.connect("clicked", hello, "")
button = builder.getButton("button2")
button.connect("clicked", hello, " again...")
button = builder.getButton("quit")
button.connect("clicked", quitApp, app)
#showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()For each builder component gintro provides a typesafe access proc like getApplicationWindow() and getButton() in this example.
Generally it is possible to use resource files merged with the executable program instead of an external XML files, we have to investigate how we can do that in Nim. And it may be possible to connect the signal handlers to handler procs from within the XML file — this is also work in progress…
GAction
GAction represents a single named action and is for GTK3 the prefered way to do user interactions. GAction works with button, menus and keyboard shortcuts.
The following example is based on
# https://wiki.gnome.org/HowDoI/GAction
# nim c gaction.nim
import gintro/[gtk, glib, gobject, gio]
proc saveCb(action: SimpleAction; v: Variant) =
echo "saveCb"
proc appActivate(app: Application) =
let window = newApplicationWindow(app)
let action = newSimpleAction("save")
discard action.connect("activate", saveCB)
window.actionMap.addAction(action)
let button = newButton()
button.label = "Save"
window.add(button)
button.setActionName("win.save")
setAccelsForAction(app, "win.save", "<Control><Shift>S")
showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()GtkApplicationWindow provides an interface to GActionMap. As the interface itself and the interface provider are defined in different modules, automatic conversion is not possible, so we have to convert the ApplicationWindow to ActionMap. (We could use a converter to do the conversion for us, but as these conversions are rare, and because gintro use no converters at all still, we use an explicit proc.) The use of cstringArray as third parameter for proc setAccelsForAction() is a bit ugly, we have to fix that later.
GMenu with GActions
The following example shows how we can define GActions and bind them to Menus, Buttons and Keyboard shortcuts. Examples for stateless actions (quit), for toggle actions (spellcheck) and for statefull actions (text justify) are provided.
Note that the following code is not a direct translation of an existing example, but a collections of informations from various sources, so it may contain bugs or not fully optimal code.
We can easily modify the above example to get the more modern look with a HeaderBar and the "Gears" MenuButtons:
While in the previous example we create only a single menu instance in proc appStartup() for all of our application windows, here we create a new menu for all of our instances in proc appActivate(). That seems to work fine, so I assume it is correct.
GMenu and GAction with GTK Builder
And here is an example from https://github.com/GNOME/gtk/blob/mainline/tests/ which uses a combination of gaction and gmenu with a GTK builder XML file for the menu description.
# nim c gaction2.nim
# https://github.com/GNOME/gtk/blob/mainline/tests/testgaction.c
# gcc -Wall gaction.c -o gaction `pkg-config --cflags --libs gtk4`
import gintro/[gtk, glib, gobject, gio]
const menuData = """
<interface>
<menu id="menuModel">
<section>
<item>
<attribute name="label">Normal Menu Item</attribute>
<attribute name="action">win.normal-menu-item</attribute>
</item>
<submenu>
<attribute name="label">Submenu</attribute>
<item>
<attribute name="label">Submenu Item</attribute>
<attribute name="action">win.submenu-item</attribute>
</item>
</submenu>
<item>
<attribute name="label">Toggle Menu Item</attribute>
<attribute name="action">win.toggle-menu-item</attribute>
</item>
</section>
<section>
<item>
<attribute name="label">Radio 1</attribute>
<attribute name="action">win.radio</attribute>
<attribute name="target">1</attribute>
</item>
<item>
<attribute name="label">Radio 2</attribute>
<attribute name="action">win.radio</attribute>
<attribute name="target">2</attribute>
</item>
<item>
<attribute name="label">Radio 3</attribute>
<attribute name="action">win.radio</attribute>
<attribute name="target">3</attribute>
</item>
</section>
</menu>
</interface>
"""
proc changeLabelButton(action: SimpleAction; v: Variant; label: Label) =
label.setLabel("Text set from button")
proc normalMenuItem(action: SimpleAction; v: Variant; label: Label) =
label.setLabel("Text set from normal menu item")
proc toggleMenuItem(action: SimpleAction; v: Variant; label: Label) =
label.setLabel("Text set from toggle menu item")
proc submenuItem(action: SimpleAction; v: Variant; label: Label) =
label.setLabel("Text set from submenu item")
proc radio(action: SimpleAction; parameter: Variant; label: Label) =
var l: uint64
let newState: Variant = newVariantString(getString(parameter, l))
let str: string = "From Radio menu item " & getString(newState, l)
label.setLabel(str)
proc bye(w: Window) =
mainQuit()
echo "Bye..."
proc main =
gtk.init()
let
window = newWindow()
box = newBox(Orientation.vertical, 12)
menubutton = newMenuButton()
button1 = newButton("Change Label Text")
label = newLabel("Initial Text")
actionGroup = newSimpleActionGroup()
window.connect("destroy", gtk.mainQuit)
#window.connect("destroy", bye)
var action = newSimpleAction("change-label-button")
discard action.connect("activate", changeLabelButton, label)
actionGroup.addAction(action)
action = newSimpleAction("normal-menu-item")
discard action.connect("activate", normalMenuItem, label)
actionGroup.addAction(action)
var v = newVariantBoolean(true)
action = newSimpleActionStateful("toggle-menu-item", nil, v)
discard action.connect("activate", toggleMenuItem, label)
actionGroup.addAction(action)
action = newSimpleAction("submenu-item")
discard action.connect("activate", subMenuItem, label)
actionGroup.addAction(action)
v = newVariantString("1")
let vt = newVariantType("s")
action = newSimpleActionStateful("radio", vt, v)
discard action.connect("activate", radio, label)
actionGroup.addAction(action)
insertActionGroup(window, "win", actionGroup)
label.setMarginTop(12)
label.setMarginBottom(12)
box.add(label)
menubutton.setHAlign(Align.center)
let builder: Builder = newBuilderFromString(menuData)
let menuModel = builder.getMenuModel("menuModel")
let menu = newMenuFromModel(menuModel)
menuButton.setPopup(menu)
box.add(menubutton)
button1.setHalign(Align.center)
button1.setActionName("win.change-label-button")
box.add(button1)
window.add(box)
window.showAll
gtk.main()
main()GSettings
GSettings provides a convenient way to permanently storing configuration data, and to bind them to properties of widgets.
You can read an introduction at https://blog.gtk.org/2017/05/01/first-steps-with-gsettings/.
For using GSettings in our own programs, we have first to create a XML file which defines names and type of each configuration entry, and additional provides default value and a description. The file name of such xml files must always end with ".gschema.xml". The following example has only one field called like-nim of type boolean (b). For a real application program we would install the configuration on our computer — unfortunately we would need root access for this. We could do it this way:
# For making gsettings available system wide one method is, as root # https://developer.gnome.org/gio/stable/glib-compile-schemas.html # echo $XDG_DATA_DIRS # /usr/share/gnome:/usr/local/share:/usr/share:/usr/share/gdm # cd /usr/local/share/glib-2.0/schemas # cp test.gschema.xml . # glib-compile-schemas . #
For testing there is an easier method available:
Create a directory and copy the xml file and the test program below into it.
Then do, as ordinary user:
glib-compile-schemas . nim c gsettings.nim GSETTINGS_SCHEMA_DIR="." ./gsettings
This is the xml file and the test program:
<schemalist>
<schema path="/org/gnome/recipes/"
id="org.gnome.Recipes">
<key type="b" name="like-nim">
<default>false</default>
<summary>I like Nim</summary>
<description>
I like or like not
the Nim programming language.
</description>
</key>
</schema>
</schemalist># gsettings.nim -- basic use of gsettings
# nim c gsettings.nim
# https://blog.gtk.org/2017/05/01/first-steps-with-gsettings/
# https://mail.gnome.org/archives/gtk-list/2016-December/msg00003.html
import gintro/[gtk, glib, gobject, gio]
# unused
proc toggle(b: CheckButton) =
echo b.active
let s = newSettings("org.gnome.Recipes")
discard s.setBoolean("like-nim", b.active)
proc appActivate(app: Application) =
let window = newApplicationWindow(app)
window.title = "GTK3, Nim and GSettings"
window.defaultSize = (200, 200)
let b = newCheckButton()
b.halign = Align.center
b.label = "I like Nim"
#b.connect("toggled", toggle) # we don't need this for plain binding!
let s = newSettings("org.gnome.Recipes")
if s.getBoolean("like-nim"):
echo "I like Nim language"
`bind`(s, "like-nim", b, "active", {SettingsBindFlag.get, SettingsBindFlag.set})
window.add(b)
showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()The command "glib-compile-schemas ." compiles all schemas in the current directory. And "GSETTINGS_SCHEMA_DIR="." ./gsettings" launches our test program with the environment variable GSETTINGS_SCHEMA_DIR pointing to the current directory, containing the compiled schema.
Note that a system tool with same name as our test program exists — that one can be used to get or set configuration data — for example you may query the current state of field "like-nim" with
gsettings --schemadir "." get org.gnome.Recipes like-nim
Or test program first creates a window with a check button. Then our settings file is opened and we print the current value of the boolean variable. After that the bind procedure binds the active property (checkmark state) of our widget to the "like-nim" entry of our settings file. The result of this binding is, that our checkmark state is automatically made persistent, that is when we terminate and restart our test program, the checkmark will have the last state again.
These bindings works for booleans, integers, floats, strings. The type of the property of the widget must be identical with the corresponding type of the entry in the settings xml file.
On Linux you may permanently set the gsetting directory by adding the statement
export GSETTINGS_SCHEMA_DIR="pathToMyProg"
to your .bashrc file — of course after replacing pathToMyProg with the actual path.
For more informations about gsettings see
Drawing with Cairo graphics library
The next example shows how we can use the cairo graphics library for drawing on a DrawingArea widget, and at the same time uses glib timeoutAdd() function to create a timer which periodically calls the drawing function to create some animations. The code is based on a recent post to the cairo mailing list and shows a sine wave which is continuously moving to the left.
| The gobject-introspection generated cairo module was only a minimal stub, because cairo library does not really support introspection. Now we are using a cairo module which is generated directly from the cairo C header files with the tool c2nim and then modified to support a high level API. |
# https://lists.cairographics.org/archives/cairo/2016-October/027791.html
# Nim version of that plain cairo animation example
import gintro/[gtk, glib, gobject, gio, cairo]
import math
const
NumPoints = 1000
Period = 100.0
proc invalidateCb(w: Widget): bool =
queueDraw(w)
return SOURCE_CONTINUE
proc sineToPoint(x, width, height: int): float =
math.sin(x.float * math.TAU / Period) * height.float * 0.5 + height.float * 0.5
proc drawingAreaDrawCb(widget: DrawingArea; context: Context): bool =
var redrawNumber {.global.} : int
let width = getAllocatedWidth(widget)
let height = getAllocatedHeight(widget)
for i in 1 ..< NumPoints:
context.lineTo(i.float , sineToPoint(i + redrawNumber, width, height))
context.stroke
inc(redrawNumber)
return true # TRUE to stop other handlers from being invoked for the event. FALSE to propagate the event further.
proc appActivate(app: Application) =
let window = newApplicationWindow(app)
window.title = "Drawing example"
window.defaultSize = (400, 400)
let drawingArea = newDrawingArea()
window.add(drawingArea)
showAll(window)
discard timeoutAdd(1000 div 60, invalidateCb, drawingArea)
connect(drawingArea, "draw", drawingAreaDrawCb)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()A simple ListView example
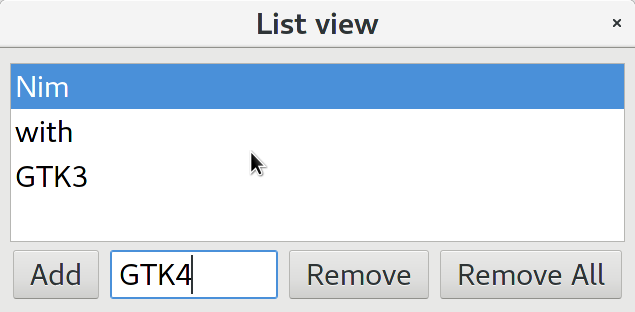
Recently someone reported about some problems porting a GTK2 application to Nim GTK3, so I will give a small example which may help using ListViews and TreeViews. These two widget types are the most complicated widget types in GTK — I can remember that I had some trouble myself when I used Ruby-GTK some years ago. As I can currently not remember details about use of ListView widgets, I decided to take an example code from zetcode.com as starting point. Of course porting is straight forward, but when I tried to compile the result I noticed some bugs and restrictions of current gintro package. Of course not really surprising, as the package is not really tested yet. I will try to fix these bugs later. First problem is, that we store a ListStore as model in our TreeView, and we need to extract that ListStore from the TreeView for some operations. But module gtk.nim offers currently only a function to extract the model itself, which is of type TreeModel. In the C code an upcast is used to get the ListStore from the retrieved TreeModel. To avoid casting in our Nim code, I have just copied the getModel() proc and modified it to return a ListStore. Second problem was, that module gio export a ListStore datatype also. To avoid prefixing all ListStore types with gtk prefix, I excluded gio.ListStore from import list. And finally a real bug: Proc newListStore() expects currently a plain pointer as last parameter, while we know that it should be the address of a list of GTypes. So we have to use an ugly cast for now. For populating the ListStore currently GValues are used. That is not very convenient, and for that we need the correct GType of our string list. In C one would use the macro G_TYPE_STRING, which is not provided by gobject-introspection. So we use typeFromName() to get the correct GType, which works fine when we know that the string name is "gchararray". Later we will provide a higher level function for this process.
I will try to give more and better explained ListView and TreeView examples later…
# http://zetcode.com/gui/gtk2/gtktreeview/
# dynamiclistview.c
import gintro/[glib, gobject, gtk]
import gintro/gio except ListStore
const
LIST_ITEM = 0
N_COLUMNS = 1
var list: TreeView
# this is copied from gtk.nim
#proc getModel*(self: TreeView): TreeModel =
# new(result)
# result.impl = gtk_tree_view_get_model(cast[ptr TreeView00](self.impl))
proc getListStore(self: TreeView): ListStore =
new(result)
result.impl = gtk_tree_view_get_model(cast[ptr TreeView00](self.impl))
proc appendItem(widget: Button; entry: Entry) =
var
val: Value
iter: TreeIter
let store = getListStore(list)
let gtype = typeFromName("gchararray")
discard gValueInit(val, gtype)
gValueSetString(val, entry.text)
store.append(iter)
store.setValue(iter, LIST_ITEM, val)
entry.text = ""
proc removeItem(widget: Button; selection: TreeSelection) =
var
ls: ListStore
iter: TreeIter
let store = getListStore(list)
if not store.getIterFirst(iter):
return
if getSelected(selection, ls, iter):
discard store.remove(iter)
proc onRemoveAll(widget: Button; selection: TreeSelection) =
var
iter: TreeIter
let store = getListStore(list)
if not store.getIterFirst(iter):
return
clear(store)
proc initList(list: TreeView) =
let renderer = newCellRendererText()
let column = newTreeViewColumn()
column.title = "List Item"
column.packStart(renderer, true)
column.addAttribute(renderer, "text", LIST_ITEM)
discard list.appendColumn(column)
let gtype = typeFromName("gchararray")
let store = newListStore(N_COLUMNS, cast[pointer]( unsafeaddr gtype)) # cast due to bug in gtk.nim
list.setModel(store)
proc appActivate(app: Application) =
let
window = newApplicationWindow(app)
sw = newScrolledWindow()
hbox = newBox(Orientation.horizontal, 5)
vbox = newBox(Orientation.vertical, 0)
add = newButton("Add")
remove = newButton("Remove")
removeAll = newButton("Remove All")
entry = newEntry()
window. title = "List view"
window.position = WindowPosition.center
window.borderWidth = 10
window.setSizeRequest(370, 270)
list = newTreeView()
sw.add(list)
sw.setPolicy(PolicyType.automatic, PolicyType.automatic)
sw.setShadowType(ShadowType.etchedIn)
list.setHeadersVisible(false)
vbox.packStart(sw, true, true, 5)
entry.setSizeRequest(120, -1)
hbox.packStart(add, false, true, 3)
hbox.packStart(entry, false, true, 3)
hbox.packStart(remove, false, true, 3)
hbox.packStart(removeAll, false, true, 3)
vbox.packStart(hbox, false, true, 3)
window.add(vbox)
initList(list)
let selection = getSelection(list)
connect(add, "clicked", listview.appendItem, entry)
connect(remove, "clicked", listview.removeItem, selection)
connect(removeAll, "clicked", listview.onRemoveAll, selection)
showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()A ListView example with CSS styling
Recently C. Eric Cashon provided this example at https://discourse.gnome.org/t/gtk-treeview-cell-color-change/1750/3
I will show his original code here too, so we can compare it better with the Nim version. We see that Nim code has currently some disadvantages still, for example we have no varargs procs implemented, so setting of properties and attributes is done using GValues, which is typesafe, but not really compact. That is not too bad, but we may consider creating macros to support a more dense, but still typesafe way similar to C’s varargs functions.
// gcc -Wall cell_color1.c -o cell_color1 `pkg-config --cflags --libs gtk+-3.0`
// https://discourse.gnome.org/t/gtk-treeview-cell-color-change/1750/4
// C. Eric Cashon
#include<gtk/gtk.h>
enum
{
ID,
PROGRAM,
COLOR1,
COLOR2,
COLUMNS
};
int main(int argc, char *argv[])
{
gtk_init(&argc, &argv);
GtkWidget *window=gtk_window_new(GTK_WINDOW_TOPLEVEL);
gtk_window_set_title(GTK_WINDOW(window), "Select Cell");
gtk_window_set_position(GTK_WINDOW(window), GTK_WIN_POS_CENTER);
gtk_window_set_default_size(GTK_WINDOW(window), 500, 500);
gtk_container_set_border_width(GTK_CONTAINER(window), 20);
g_signal_connect(window, "destroy", G_CALLBACK(gtk_main_quit), NULL);
GtkTreeIter iter;
GtkListStore *store=gtk_list_store_new(COLUMNS, G_TYPE_UINT, G_TYPE_STRING, G_TYPE_STRING, G_TYPE_STRING);
gtk_list_store_append(store, &iter);
gtk_list_store_set(store, &iter, ID, 0, PROGRAM, "Gedit", COLOR1, "DarkCyan", COLOR2, "cyan", -1);
gtk_list_store_append(store, &iter);
gtk_list_store_set(store, &iter, ID, 1, PROGRAM, "Gimp", COLOR1, "LightSlateGray", COLOR2, "cyan", -1);
gtk_list_store_append(store, &iter);
gtk_list_store_set(store, &iter, ID, 2, PROGRAM, "Inkscape", COLOR1, "DarkCyan", COLOR2, "cyan", -1);
gtk_list_store_append(store, &iter);
gtk_list_store_set(store, &iter, ID, 3, PROGRAM, "Firefox", COLOR1, "LightSlateGray", COLOR2, "cyan", -1);
gtk_list_store_append(store, &iter);
gtk_list_store_set(store, &iter, ID, 4, PROGRAM, "Calculator", COLOR1, "DarkCyan", COLOR2, "cyan", -1);
gtk_list_store_append(store, &iter);
gtk_list_store_set(store, &iter, ID, 5, PROGRAM, "Devhelp", COLOR1, "LightSlateGray", COLOR2, "cyan", -1);
GtkWidget *tree=gtk_tree_view_new_with_model(GTK_TREE_MODEL(store));
gtk_widget_set_hexpand(tree, TRUE);
gtk_widget_set_vexpand(tree, TRUE);
g_object_set(tree, "activate-on-single-click", TRUE, NULL);
GtkTreeSelection *selection=gtk_tree_view_get_selection(GTK_TREE_VIEW(tree));
gtk_tree_selection_set_mode(selection, GTK_SELECTION_SINGLE);
GtkCellRenderer *renderer1=gtk_cell_renderer_text_new();
g_object_set(renderer1, "editable", FALSE, NULL);
GtkCellRenderer *renderer2=gtk_cell_renderer_text_new();
g_object_set(renderer2, "editable", TRUE, NULL);
//Bind the COLOR column to the "cell-background" property.
GtkTreeViewColumn *column1=gtk_tree_view_column_new_with_attributes("ID", renderer1, "text", ID, "cell-background", COLOR1, NULL);
gtk_tree_view_append_column(GTK_TREE_VIEW(tree), column1);
GtkTreeViewColumn *column2 = gtk_tree_view_column_new_with_attributes("Program", renderer2, "text", PROGRAM, "cell-background", COLOR2, NULL);
gtk_tree_view_append_column(GTK_TREE_VIEW(tree), column2);
GtkWidget *grid=gtk_grid_new();
gtk_grid_attach(GTK_GRID(grid), tree, 0, 0, 1, 1);
gtk_container_add(GTK_CONTAINER(window), grid);
gchar *css_string=g_strdup("treeview{background-color: rgba(0,255,255,1.0); font-size:30pt} treeview:selected{background-color: rgba(255,255,0,1.0); color: rgba(0,0,255,1.0);}");
GError *css_error=NULL;
GtkCssProvider *provider=gtk_css_provider_new();
gtk_css_provider_load_from_data(provider, css_string, -1, &css_error);
gtk_style_context_add_provider_for_screen(gdk_screen_get_default(), GTK_STYLE_PROVIDER(provider), GTK_STYLE_PROVIDER_PRIORITY_APPLICATION);
if(css_error!=NULL)
{
g_print("CSS loader error %s\n", css_error->message);
g_error_free(css_error);
}
g_object_unref(provider);
g_free(css_string);
gtk_widget_show_all(window);
gtk_main();
return 0;
}And this is the Nim version, created with c2nim and some manual tuning:
# nim c css_colored_listview.nim
import gintro/[gtk, glib, gobject]
import gintro/gdk except Window # there is a problem with gdk.Window -- we have to investigate!
const # maybe we should use Nim's enum here?
Id = 0
Program = 1
Color1 = 2
Color2 = 3
Columns = 4
proc bye(w: Window) =
mainQuit()
echo "Bye..."
proc toStringVal(s: string): Value =
let gtype = typeFromName("gchararray")
discard init(result, gtype)
setString(result, s)
proc toUIntVal(i: int): Value =
let gtype = typeFromName("guint")
discard init(result, gtype)
setUint(result, i)
proc toBoolVal(b: bool): Value =
let gtype = typeFromName("gboolean")
discard init(result, gtype)
setBoolean(result, b)
# we need the following two procs for now -- later we will not use that ugly cast...
proc typeTest(o: gobject.Object; s: string): bool =
let gt = g_type_from_name(s)
return g_type_check_instance_is_a(cast[ptr TypeInstance00](o.impl), gt).toBool
proc listStore(o: gobject.Object): gtk.ListStore =
assert(typeTest(o, "GtkListStore"))
cast[gtk.ListStore](o)
proc updateRow(renderer: CellRendererText; path: cstring; newText: cstring; tree: TreeView) =
var iter: TreeIter
var value: Value
let gtype = typeFromName("gchararray")
discard init(value, gtype)
let store = listStore(tree.getModel())
value.setString(newText)
let treePath = newTreePathFromString(path)
discard store.getIter(iter, treePath)
store.setValue(iter, 1, value)
# we use the old gtk style with init() as is used in the C original -- maybe better use modern app sytle
proc main() =
gtk.init()
let window = newWindow()
window.title = "Select Cell"
window.position = WindowPosition.center
window.defaultSize = (500, 500)
window.borderWidth = 20
connect(window, "destroy", bye)
var iter: TreeIter
var h = [typeFromName("guint"), typeFromName("gchararray"), typeFromName("gchararray"),
typeFromName("gchararray")]
var store = newListStore(Columns, cast[pointer]( unsafeaddr h)) # cast is ugly, we should fix it in bindings.
let progNames = ["Gedit", "Gimp", "Inkscape", "Firefox", "Calculator", "Devhelp"]
for i, n in progNames:
store.append(iter) # currently we have to use setValue() as there is no varargs proc as in C original
store.setValue(iter, Id, toUIntVal(i))
store.setValue(iter, Program, toStringVal(n))
store.setValue(iter, Color1, toStringVal(if (i and 1) != 0: "LightSlateGray" else: "DarkCyan"))
store.setValue(iter, Color2, toStringVal("cyan"))
var tree = newTreeViewWithModel(store)
tree.setHexpand
tree.setVexpand
setProperty(tree, "activate-on-single-click", toBoolVal(true))
var selection = tree.getSelection()
selection.setMode(SelectionMode.single)
var renderer1 = newCellRendererText()
setProperty(renderer1, "editable", toBoolVal(false))
var renderer2 = newCellRendererText()
setProperty(renderer2, "editable", toBoolVal(true))
connect(renderer2, "edited", updateRow, tree)
## Bind the Color column to the "cell-background" property.
var column1 = newTreeViewColumn()
column1.setTitle("ID")
column1.packStart(renderer1, true)
column1.addAttribute(renderer1, "text", Id)
column1.addAttribute(renderer1, "cell-background", Color1)
discard tree.appendColumn(column1)
var column2 = newTreeViewColumn()
column1.setTitle("Program")
column1.packStart(renderer2, true)
column1.addAttribute(renderer2, "text", Program)
column1.addAttribute(renderer2, "cell-background", Color2)
discard tree.appendColumn(column2)
var grid = newGrid() # only one occupied cell makes no sense -- but so we can add more widgets later
grid.attach(tree, 0, 0, 1, 1)
window.add(grid)
const cssString = # note: big font selected intentionally
"""treeview{background-color: rgba(0,255,255,1.0); font-size:30pt} treeview:selected{background-color:
rgba(255,255,0,1.0); color: rgba(0,0,255,1.0);}"""
var provider = newCssProvider()
discard provider.loadFromData(cssString)
addProviderForScreen(getDefaultScreen(), provider, STYLE_PROVIDER_PRIORITY_APPLICATION)
window.showAll
gtk.main()
main()When you compile with nim c -d:release -d:danger --passC:-flto css_colored_listview.nim
you will get an executable size of 80k, which is big compared with the 20k of the C version, but
not too bad. You may note that I have added the updateRow() proc, which is necessary to
make editing the program name entries permanent. That proc needs cstring parametes, which
may be surprising, as we generally use Nim strings. Not a big problem, maybe intended, we may have to
check the connect() macro in gimpl.nim.
And one more Listview example — with custom cairo drawing
This example is again a Nim version of a C example from C. Eric Cashon provided at https://discourse.gnome.org/t/gtk-how-to-draw-on-top-of-gtktreeview/1783/2.
It draws an rectangular frame on a selected listview cell. For that to work connectAfter() is used to ensure that the custom cairo drawing occurs after the widget is drawn by GTK.
# nim c overlayTree1.nim
import gintro/[gtk, gdk, glib, gobject, cairo]
import strformat
from strutils import parseInt
const
Id = 0
Program = 1
Color = 2
Color2 = 3
Columns = 4
var
rowG = 0
columnG = 1
proc bye(w: gtk.Window) =
mainQuit()
echo "Bye..."
proc toStringVal(s: string): Value =
let gtype = typeFromName("gchararray")
discard init(result, gtype)
setString(result, s)
proc toUIntVal(i: int): Value =
let gtype = typeFromName("guint")
discard init(result, gtype)
setUint(result, i)
proc toBoolVal(b: bool): Value =
let gtype = typeFromName("gboolean")
discard init(result, gtype)
setBoolean(result, b)
proc selectCell(treeView: TreeView; path: TreePath; column: TreeViewColumn) =
let str = toString(path)
echo fmt"{str} {getTitle(column)}"
rowG = parseInt(str)
queueDraw(treeView)
proc drawRectangle(overlay: Overlay; cr: cairo.Context; treeView: TreeView): bool =
echo fmt"Draw Rectangle {rowG} {columnG}"
let path = newTreePathFromIndices(@[rowG.int32])
echo path.toString
let column = getColumn(treeView, columnG)
var rect: gdk.Rectangle
var x, y: int
treeView.convertBinWindowToWidgetCoords(0, 0, x, y)
cr.save
cr.translate(x.float, y.float)
cr.setLineWidth(2)
cr.setSource(0, 0, 0, 1)
treeView.getCellArea(path, column, rect)
cr.rectangle(rect.x.float + 1, rect.y.float + 1, rect.width.float - 1, rect.height.float - 1)
cr.stroke
cr.restore
return EVENT_PROPAGATE # false
proc main =
gtk.init()
let window = newWindow()
window.setTitle("Overlay Tree")
window.setPosition(WindowPosition.center)
window.setDefaultSize(500, 500)
window.setBorderWidth(20)
window.connect("destroy", bye)
var iter: TreeIter
let h = [typeFromName("guint"), typeFromName("gchararray"), typeFromName("gchararray"),
typeFromName("gchararray")]
let store = newListStore(Columns, cast[pointer](unsafeaddr h)) # cast is ugly, we should fix it in bindings.
let progNames = ["Gedit", "Gimp", "Inkscape", "Firefox", "Calculator", "Devhelp"]
for i, n in progNames:
store.append(iter) # currently we have to use setValue() as there is no varargs proc as in C original
store.setValue(iter, Id, toUIntVal(i))
store.setValue(iter, Program, toStringVal(n))
store.setValue(iter, Color, toStringVal("SpringGreen"))
store.setValue(iter, Color2, toStringVal("cyan"))
let tree = newTreeViewWithModel(store)
tree.setHexpand
tree.setVexpand
tree.setProperty("activate-on-single-click", toBoolVal(true))
let selection = tree.getSelection
selection.setMode(SelectionMode.single)
let renderer1 = newCellRendererText()
renderer1.setProperty("editable", toBoolVal(false))
let renderer2 = newCellRendererText()
renderer2.setProperty("editable", toBoolVal(true))
tree.connect("row-activated", selectCell)
## Bind the COLOR column to the "cell-background" property.
let column1 = newTreeViewColumn()
column1.setTitle("ID")
column1.packStart(renderer1, true)
column1.addAttribute(renderer1, "text", Id)
column1.addAttribute(renderer1, "cell-background", Color)
discard tree.appendColumn(column1)
let column2 = newTreeViewColumn()
column2.setTitle("Program")
column2.packStart(renderer2, true)
column2.addAttribute(renderer2, "text", Program)
column2.addAttribute(renderer2, "cell-background", Color2)
discard tree.appendColumn(column2)
## For drawing the outline of the cell.
let overlay = newOverlay()
overlay.setHexpand
overlay.setVexpand
overlay.setAppPaintable
overlay.addOverlay(tree)
overlay.setOverlayPassThrough(tree, true)
overlay.connectAfter("draw", drawRectangle, tree)
let grid = newGrid()
grid.attach(overlay, 0, 0, 1, 1)
window.add(grid)
const cssString =
"""treeview{background-color: rgba(0,255,255,1.0);
font-size:30pt} treeview:selected{background-color:rgba(0,255,255,1.0);
color: rgba(0,0,255,1.0);}"""
let provider = newCssProvider()
discard provider.loadFromData(cssString)
getDefaultScreen().addProviderForScreen(provider, STYLE_PROVIDER_PRIORITY_APPLICATION)
window.showAll
gtk.main()
main() # 123 linesA Listview example using a CellDataFunction
This example shows how a CellDataFunction can be used to customize cells of a Tree- or Listview.
# This example shows how to apply a CellDataFunc to a GtkTreeView
# C example code was provided by A.Krause in chapter 8 of his book
import gintro/[gtk, gobject, glib]
const
Color = 0
Columns = 1
clr = ["00", "33", "66", "99", "CC", "FF"]
proc bye(w: Window) =
mainQuit()
echo "Bye..."
proc toStringVal(s: string): Value =
let gtype = gStringGetType() # typeFromName("gchararray")
discard init(result, gtype)
setString(result, s)
proc toBoolVal(b: bool): Value =
let gtype = gBooleanGetType() # typeFromName("gboolean")
discard init(result, gtype)
setBoolean(result, b)
# our Nim function
proc cellDataFuncN(column: TreeViewColumn; renderer: CellRenderer;
model: TreeModel; iter: TreeIter, data: TreeViewColumn) =
## Get the color string stored by the column and make it the foreground color.
# for testing that optional args work, we pass a TreeViewColumn and echo its title
echo data.title
var val: Value
model.getValue(iter, Color, val)
let text = val.getString
val.unset # is this necessary?
setProperty(renderer, "foreground", toStringVal("#FFFFFF"))
setProperty(renderer, "foreground-set", toBoolVal(true))
setProperty(renderer, "background", toStringVal(text))
setProperty(renderer, "background-set", toBoolVal(true))
setProperty(renderer, "text", toStringVal(text))
## Add three columns to the GtkTreeView. All three of the columns will be
## displayed as text, although one is a gboolean value and another is
## an integer.
proc setupTreeView(treeview: TreeView) =
let renderer = gtk.newCellRendererText()
let column = newTreeViewColumn()
column.title = "Standard Colors"
column.packStart(renderer, expand = true)
column.addAttribute(renderer, "text", Color)
discard treeview.appendColumn(column)
column.setCellDataFunc(renderer, cellDataFuncN, column)
column.setCellDataFunc(renderer) # test unsetting!
column.setCellDataFunc(renderer, nil)
column.unsetCellDataFunc(renderer)
column.setCellDataFunc(renderer, cellDataFuncN, column)
proc main =
var iter: TreeIter
gtk.init()
let window = newWindow()
window.setTitle("Color List")
window.setBorderWidth(10)
window.setSizeRequest(250, 175)
window.connect("destroy", bye)
let treeview = newTreeView()
setupTreeView(treeview)
let gtype = typeFromName("gchararray")
let store = newListStore(Columns, cast[pointer](unsafeaddr gtype)) # ugly cast
## Add all of the products to the GtkListStore.
for i in 0 ..< 6:
for j in 0 ..< 6:
for k in 0 ..< 6:
let color: string = "#" & clr[i] & clr[j] & clr[k]
store.append(iter)
store.setValue(iter, Color, toStringVal(color))
treeView.setModel(store)
let scrolledWin = newScrolledWindow(nil, nil)
scrolledWin.setPolicy(PolicyType.automatic, PolicyType.automatic)
scrolledWin.add(treeview)
window.add(scrolledWin)
window.showAll
gtk.main()
main()A more advanced example for cairo drawing with zooming, panning, scrolling
The following code is a plain Nim version of a drawing demo which I wrote some years ago in Ruby (http://ssalewski.de/PetEd-Demo.html.en). Cairo surface is currently manually freed, because GC may have a too large delay.
You can resize the window and zoom in with the mouse wheel. When zoomed in scroll bars appear. You can hold the middle mouse button pressed while moving the mouse for panning, and you can press left mouse button and move the mouse to first draw a selection rectangle and zoom into it when releasing the mouse button.
In the examples directory there is also a simplified version called simpledrawingarea.nim which does all
the drawings in the draw callback, without using a buffering surface. This is generally preferable for
plain applications.
# Plain demo for zooming, panning, scrolling with GTK DrawingArea
# (c) S. Salewski, 21-DEC-2010 (initial Ruby version)
# Nim version April 2019
# License MIT
# This version of the demo program uses a separate proc paint()
# which allocates a custom surface for buffered drawing.
# That may be not really necessary, for simple drawings doing all
# the drawing in the "draw" call back is easier and faster. But for
# more complicated drawing operations, for example when using a
# background grid, which is a bit larger than the window size and
# is reused when scrolling, a custom surface may be useful.
# And finally that custom surface and custom cairo context is an
# important test for the language bindings.
# https://discourse.gnome.org/t/problem-with-gtkscrollbar-gtk-window-resize-and-gtk-adjustment-set-value/1081
import gintro/[gtk, gdk, glib, gobject, gio, cairo]
const
ZoomFactorMouseWheel = 1.1
ZoomFactorSelectMax = 10 # ignore zooming in tiny selection
ZoomNearMousepointer = true # mouse wheel zooming -- to mouse-pointer or center
SelectRectCol = [0.0, 0, 1, 0.5] # blue with transparency
discard """
Zooming, scrolling, panning...
|-------------------------|
|<-------- A ------------>|
| |
| |---------------| |
| | <---- a ----->| |
| | visible | |
| |---------------| |
| |
| |
|-------------------------|
a is the visible, zoomed in area == darea.allocatedWidth
A is the total data range
A/a == userZoom >= 1
For horizontal adjustment we use
hadjustment.setUpper(darea.allocatedWidth * userZoom) == A
hadjustment.setPageSize(darea.allocatedWidth) == a
So hadjustment.value == left side of visible area
Initially, we set userZoom = 1, scale our data to fit into darea.allocatedWidth
and translate the origin of our data to (0, 0)
Zooming: Mouse wheel or selecting a rectangle with left mouse button pressed
Scrolling: Scrollbars
Panning: Moving mouse while middle mouse button pressed
"""
# drawing area and scroll bars in 2x2 grid (PDA == Plain Drawing Area)
type
PosAdj = ref object of Adjustment
handlerID: uint64
proc newPosAdj: PosAdj =
initAdjustment(result, 0, 0, 1, 1, 10, 1)
type
PDA_Data* = object
draw*: proc (cr: Context)
extents*: proc (): tuple[x, y, w, h: float]
windowSize*: tuple[w, h: int]
type
PDA = ref object of Grid
zoomNearMousepointer: bool
selecting: bool
userZoom: float
surf: Surface
pattern: Pattern
cr: cairo.Context
darea: DrawingArea
hadjustment: PosAdj
vadjustment: PosAdj
hscrollbar: Scrollbar
vscrollbar: Scrollbar
fullScale: float
dataX: float
dataY: float
dataWidth: float
dataHeight: float
lastButtonDownPosX: float
lastButtonDownPosY: float
lastMousePosX: float
lastMousePosY: float
zoomRectX1: float
zoomRectY1: float
oldSizeX: int
oldSizeY: int
drawWorld: proc (cr: Context)
extents: proc (): tuple[x, y, w, h: float]
proc drawingAreaDrawCb(darea: DrawingArea; cr: Context; this: PDA): bool =
if this.pattern.isNil: return
cr.setSource(this.pattern)
cr.paint
if this.selecting:
cr.rectangle(this.lastButtonDownPosX, this.lastButtonDownPosY,
this.zoomRectX1 - this.lastButtonDownPosX, this.zoomRectY1 - this.lastButtonDownPosY)
cr.setSource(0, 0, 1, 0.5) # SELECT_RECT_COL) # 0, 0, 1, 0.5
cr.fillPreserve
cr.setSource(0, 0, 0)
cr.setLineWidth(2)
cr.stroke
return gdk.EVENT_STOP # EVENT_PROPAGATE
#return true # TRUE to stop other handlers from being invoked for the event. FALSE to propagate the event further.
# clamp to correct values, 0 <= value <= (adj.upper - adj.pageSize), block calling onAdjustmentEvent()
proc updateVal(adj: PosAdj; d: float) =
adj.signalHandlerBlock(adj.handlerID)
adj.setValue(max(0.0, min(adj.value + d, adj.upper - adj.pageSize)))
adj.signalHandlerUnblock(adj.handlerID)
proc updateAdjustments(this: PDA; dx, dy: float) =
this.hadjustment.setUpper(this.darea.allocatedWidth.float * this.userZoom)
this.vadjustment.setUpper(this.darea.allocatedHeight.float * this.userZoom)
this.hadjustment.setPageSize(this.darea.allocatedWidth.float)
this.vadjustment.setPageSize(this.darea.allocatedHeight.float)
updateVal(this.hadjustment, dx)
updateVal(this.vadjustment, dy)
proc paint(this: PDA) =
# echo "paint"
this.cr.save
this.cr.translate(this.hadjustment.upper * 0.5 - this.hadjustment.value, # our origin is the center
this.vadjustment.upper * 0.5 - this.vadjustment.value)
this.cr.scale(this.fullScale * this.userZoom, this.fullScale * this.userZoom)
this.cr.translate(-this.dataX - this.dataWidth * 0.5, -this.dataY - this.dataHeight * 0.5)
this.drawWorld(this.cr) # call the user provided drawing function
this.cr.restore
proc dareaConfigureCallback(darea: DrawingArea; event: EventConfigure; this: PDA): bool =
(this.dataX, this.dataY, this.dataWidth,
this.dataHeight) = this.extents() # query user defined size
this.fullScale = min(this.darea.allocatedWidth.float / this.dataWidth,
this.darea.allocatedHeight.float / this.dataHeight)
if this.surf != nil:
destroy(this.surf) # manually destroy surface -- GC would do it for us, but GC is slow...
this.surf = this.darea.window.createSimilarSurface(Content.color,
this.darea.allocatedWidth, this.darea.allocatedHeight)
if this.pattern != nil:
patternDestroy(this.pattern)
if this.cr != nil:
destroy(this.cr)
this.pattern = patternCreateForSurface(this.surf) # pattern now owns the surface!
this.cr = newContext(this.surf) # this function references target!
this.paint
return gdk.EVENT_STOP
proc hscrollbarSizeAllocateCallback(s: Scrollbar; r: gdk.Rectangle; pda: PDA) =
pda.hadjustment.setUpper(r.width.float * pda.userZoom)
pda.hadjustment.setPageSize(r.width.float)
if pda.oldSizeX != 0: # this fix is not exact, as fullScale can ...
updateVal(pda.hadjustment, (r.width - pda.oldSizeX).float * 0.5)
pda.oldSizeX = r.width
proc vscrollbarSizeAllocateCallback(s: Scrollbar; r: gdk.Rectangle; pda: PDA) =
pda.vadjustment.setUpper(r.height.float * pda.userZoom)
pda.vadjustment.setPageSize(r.height.float)
if pda.oldSizeY != 0: # ... change when window is rezized. But it's good enough!
updateVal(pda.vadjustment, (r.height - pda.oldSizeY).float * 0.5)
pda.oldSizeY = r.height
proc updateAdjustmentsAndPaint(this: PDA; dx, dy: float) =
this.updateAdjustments(dx, dy)
this.paint
this.darea.queueDrawArea(0, 0, this.darea.allocatedWidth, this.darea.allocatedHeight)
# event coordinates to user space
proc getUserCoordinates(this: PDA; eventX, eventY: float): (float, float) =
((eventX - this.hadjustment.upper * 0.5 + this.hadjustment.value) / (
this.fullScale * this.userZoom) + this.dataX + this.dataWidth * 0.5,
(eventY - this.vadjustment.upper * 0.5 + this.vadjustment.value) / (
this.fullScale * this.userZoom) + this.dataY + this.dataHeight * 0.5)
proc onMotion(darea: DrawingArea; event: EventMotion; this: PDA): bool =
let state = getState(event)
let (x, y) = event.getCoords
if state.contains(button1): # selecting
this.selecting = true
this.zoomRectX1 = x
this.zoomRectY1 = y
this.darea.queueDrawArea(0, 0, this.darea.allocatedWidth, this.darea.allocatedHeight)
elif button2 in state: # panning
this.updateAdjustmentsAndPaint(this.lastMousePosX - x, this.lastMousePosY - y)
else:
return gdk.EVENT_PROPAGATE
this.lastMousePosX = x
this.lastMousePosY = y
return gdk.EVENT_STOP
#event.request # request more motion events ?
# zooming with mouse wheel -- data near mouse pointer should not move if possible!
# hadjustment.value + event.x is the position in our zoomed_in world, (userZoom / z0 - 1)
# is the relative movement caused by zooming
# In other words, this is the delta-move d of a point at position P from zooming:
# d = newPos - P = P * scale - P = P * (z/z0) - P = P * (z/z0 - 1). We have to compensate for this d.
proc scrollEvent(darea: DrawingArea; event: EventScroll; this: PDA): bool =
let z0 = this.userZoom
case getScrollDirection(event)
of ScrollDirection.up:
this.userZoom *= ZoomFactorMouseWheel
of ScrollDirection.down:
this.userZoom /= ZoomFactorMouseWheel
if this.userZoom < 1:
this.userZoom = 1
else:
return gdk.EVENT_PROPAGATE
if this.zoomNearMousepointer:
let (x, y) = event.getCoords
this.updateAdjustmentsAndPaint((this.hadjustment.value + x) * (this.userZoom / z0 - 1),
(this.vadjustment.value + y) * (this.userZoom / z0 - 1))
else: # zoom to center
this.updateAdjustmentsAndPaint((this.hadjustment.value +
this.darea.allocatedWidth.float * 0.5) * (this.userZoom / z0 - 1),
(this.vadjustment.value + this.darea.allocatedHeight.float * 0.5) * (this.userZoom / z0 - 1))
return gdk.EVENT_STOP
proc buttonPressEvent(darea: DrawingArea; event: EventButton; this: PDA): bool =
var (x, y) = event.getCoords
this.lastMousePosX = x
this.lastMousePosY = y
this.lastButtonDownPosX = x
this.lastButtonDownPosY = y
echo "buttonPressEvent", x, " ", y
(x, y) = this.getUserCoordinates(x, y)
echo "User coordinates: ", x, ' ', y, "\n" # to verify getUserCoordinates()
return gdk.EVENT_STOP
# zoom into selected rectangle and center it
# math: we first center the selection rectangle, and then compensate for translation due to scale
proc buttonReleaseEvent(darea: DrawingArea; event: EventButton; this: PDA): bool =
let (x, y) = event.getCoords
let b = getButton(event)
if b == 1:
this.selecting = false
let z1 = min(this.darea.allocatedWidth.float / (this.lastButtonDownPosX - x).abs,
this.darea.allocatedHeight.float / (this.lastButtonDownPosY - y).abs)
if z1 < ZoomFactorSelectMax: # else selection rectangle will persist, we may output a message...
this.userZoom *= z1
this.updateAdjustmentsAndPaint(
((x + this.lastButtonDownPosX) * z1 - this.darea.allocatedWidth.float) * 0.5 + this.hadjustment.value * (z1 - 1),
((y + this.lastButtonDownPosY) * z1 - this.darea.allocatedHeight.float) * 0.5 + this.vadjustment.value * (z1 - 1))
return gdk.EVENT_STOP
return gdk.EVENT_PROPAGATE
proc onAdjustmentEvent(this: PosAdj; pda: PDA) =
pda.paint
pda.darea.queueDrawArea(0, 0, pda.darea.allocatedWidth, pda.darea.allocatedHeight)
proc newPDA: PDA =
initGrid(result)
let da = newDrawingArea()
result.darea = da
da.setHExpand
da.setVExpand
da.connect("draw", drawingAreaDrawCb, result)
da.connect("configure-event", dareaConfigureCallback, result)
da.addEvents({EventFlag.buttonPress, EventFlag.buttonRelease,
EventFlag.scroll, button1Motion, button2Motion, pointerMotionHint})
da.connect("motion-notify-event", onMotion, result)
da.connect("scroll_event", scrollEvent, result)
da.connect("button_press_event", buttonPressEvent, result)
da.connect("button_release_event", buttonReleaseEvent, result)
result.zoomNearMousepointer = ZoomNearMousepointer # mouse wheel zooming
result.userZoom = 1.0
result.hadjustment = newPosAdj()
result.hadjustment.handlerID = result.hadjustment.connect("value-changed", onAdjustmentEvent, result)
result.vadjustment = newPosAdj()
result.vadjustment.handlerID = result.vadjustment.connect("value-changed", onAdjustmentEvent, result)
result.hscrollbar = newScrollbar(Orientation.horizontal, result.hadjustment)
result.vscrollbar = newScrollbar(Orientation.vertical, result.vadjustment)
result.hscrollbar.setHExpand
result.vscrollbar.setVExpand
result.hscrollbar.connect("size-allocate", hscrollbarSizeAllocateCallback, result)
result.vscrollbar.connect("size-allocate", vscrollbarSizeAllocateCallback, result)
result.attach(result.darea, 0, 0, 1, 1)
result.attach(result.vscrollbar, 1, 0, 1, 1)
result.attach(result.hscrollbar, 0, 1, 1, 1)
proc appStartup(app: Application) =
echo "appStartup"
proc appActivate(app: Application; initData: PDA_Data) =
let window = newApplicationWindow(app)
window.title = "Drawing example"
# window.defaultSize = initData.windowSize
window.defaultSize = (initData.windowSize[0], initData.windowSize[1])
let pda = newPDA()
pda.drawWorld = initData.draw
pda.extents = initData.extents
window.add(pda)
showAll(window)
proc newDisplay*(initData: PDA_Data) =
let app = newApplication("org.gtk.example")
connect(app, "startup", appStartup)
connect(app, "activate", appActivate, initData)
discard run(app)
when isMainModule:
const # arbitrary locations for our data
DataX = 150.0
DataY = 250.0
DataWidth = 200.0
DataHeight = 120.0
# we need two user defined functions -- one gives the extent of the graphics,
# and the other does the cairo drawing using a cairo context.
# bounding box of user data -- x, y, w, h -- top left corner, width, height
proc worldExtents(): (float, float, float, float) =
(DataX, DataY, DataWidth, DataHeight) # current extents of our user world
# draw to cairo context
proc drawWorld(cr: cairo.Context) =
cr.setSource(1, 1, 1)
cr.paint
cr.setSource(0, 0, 0)
cr.setLineWidth(2)
var i = 0.0
while min(DataWidth - 2 * i, DataHeight - 2 * i) > 0:
cr.rectangle(DataX + i, DataY + i, DataWidth - 2 * i, DataHeight - 2 * i)
i += 10
cr.stroke
proc test =
let data = PDA_Data(draw: drawWorld, extents: worldExtents, windowSize: (800, 600))
newDisplay(data)
test() # 337 linesWe can use this module as a library easily and get this simple drawing tool with full zoom and scroll support:
import gintro/cairo
import drawingarea
from math import PI
proc extents(): (float, float, float, float) =
(0.0, 0.0, 100.0, 100.0) # ugly float literals
# draw to cairo context
proc draw(cr: cairo.Context) =
cr.setSource(1, 1, 1) # set background color and paint
cr.paint
cr.setSource(0, 0, 0) # forground color
cr.arc(20, 30, 10, 0, 5) # nearly a circle
cr.newSubPath # do not join the two arcs
cr.arc(70, 60, 20, 0, math.PI)
cr.stroke # finally do it
proc main =
var data: PDA_Data
data.draw = draw
data.extents = extents
data.windowSize = (800, 600)
newDisplay(data)
main()One more cairo example
Recently Mr. C. Eric Cashon provided an example code for working with a large bitmap image. His example writes the image to disk, loads it again and displays the image allowing zooming and translation. As examples are rare in these days, and that example is not to large, I used c2nim to convert it to Nim. Below is the code with a few manually fixes. Note, the current shipped cairo.nim module contains an assert statement, which prevents running this example. If you really intent running this code, you will have to fix that single line in cairo.nim. I have to do some more fixes in the cairo module and may ship a new version eventually. This example is really low level, as alloc() is used directly.
# https://discourse.gnome.org/t/proper-zoom-pan-image-approach-for-large-images/1497/6
# Nim version of the C example of C. Eric Cashon
import gintro/[gtk, gobject, glib, cairo]
from math import TAU
import strutils
const
Width = 5000
Height = 5000
CFormat = cairo.Format.argb32
var
Key: cairo.UserDataKey
translateX: float
translateY: float
scale = 1.0
## Store data from file.
bigSurfaceData*: ptr cuchar = nil
proc translateXSpinChanged(spinButton: SpinButton; data: DrawingArea) =
translateX = spinButton.value
data.queueDraw
proc translateYSpinChanged(spinButton: SpinButton; data: DrawingArea) =
translateY = spinButton.getValue
data.queueDraw
proc scaleSpinChanged(spinButton: SpinButton; data: DrawingArea) =
scale = spinButton.value
data.queueDraw
proc saveBigSurface =
## Use gdk_cairo_surface_create_from_pixbuf() to read in a pixbuf. Try a test surface here.
let bigSurface = imageSurfaceCreate(CFormat, Width, Height)
let cr = newContext(bigSurface)
## Paint the background.
cr.setSource(1, 1, 1)
cr.paint
## Draw a circle.
cr.setSource(0, 0, 1)
cr.arc(250, 250, 50, 0, math.TAU)
cr.fill
## Draw some test grid lines.
cr.setSource(0, 1, 0)
for i in countup(0, 4900, 100):
cr.moveTo(0, i.float)
cr.lineTo(5000, i.float)
cr.stroke
for i in countup(0, 4900, 100):
cr.moveTo(i.float, 0)
cr.lineTo(i.float, 5000)
cr.stroke
cr.setSource(0, 0, 1)
cr.setLineWidth(10)
for i in 0 ..< 10:
cr.moveTo(0, i.float * 500.0)
cr.lineTo(5000, i.float * 500.0)
cr.stroke
for i in 0 ..< 10:
cr.moveTo(i.float * 500.0, 0)
cr.lineTo(i.float * 500.0, 5000)
cr.stroke
## Outside box.
cr.setLineWidth(20)
cr.setSource(1, 0, 1)
cr.rectangle(0, 0, 5000, 5000)
cr.stroke
## Save surface data to file.
let f: File = open("big_surface.s", fmWrite)
let p: ptr cuchar = cairo_image_surface_get_data(bigSurface.impl)
let len = writeBuffer(f, p, cairo_format_stride_for_width(CFormat, Width) * Height)
echo("write $1\n" % $len)
close(f)
proc myDealloc(data: pointer) {.cdecl.} =
system.dealloc(data)
proc getBigSurface(): Surface =
let f: File = open("big_surface.s", fmRead)
# setFilePos(f, 0)
# https://www.cairographics.org/manual/cairo-Image-Surfaces.html#cairo-format-stride-for-width
let stride = cairo_format_stride_for_width(CFormat, Width)
bigSurfaceData = cast[ptr cuchar](malloc((stride * Height).uint64))
var len = readBuffer(f, bigSurfaceData, stride * Height)
echo("read $1" % $len)
close(f)
let bigSurface: Surface = new Surface # this is a temporary fix, we will support this later in cairo modul
bigSurface.impl = cairo_image_surface_create_for_data(bigSurfaceData, CFormat, Width, Height, stride)
discard setUserData(bigSurface, addr(Key), bigSurfaceData, myDealloc) # automatic deallocation
# flush(bigSurface)
echo("open $1" % bigSurface.status.statusToString)
return bigSurface
proc daDrawing*(da: DrawingArea; cr: Context; bigSurface: Surface): bool =
var
width = da.getAllocatedWidth.float
height = da.getAllocatedHeight.float
originX = translateX
originY = translateY
## Some constraints.
if translateX > 5000.0 - width:
originX = 5000.0 - width / scale
if translateY > 5000.0 - height:
originY = 5000.0 - height / scale
cr.setSource(0, 0, 0)
cr.paint
## Partition the big surface.
var littleSurface: Surface = cairo.surfaceCreateForRectangle(bigSurface,
originX, originY, width / scale, height / scale)
cr.scale(scale, scale)
cr.setSourceSurface(littleSurface, 0, 0)
setFilter(getSource(cr), cairo.Filter.bilinear)
cr.paint
return true
proc bye(w: Window) =
mainQuit()
echo "Bye..."
proc main =
gtk.init()
let window = newWindow()
window.setTitle("Big Surface2")
window.setDefaultSize(500, 500)
window.setPosition(gtk.WindowPosition.center)
window.connect("destroy", bye)
## Get a test surface.
saveBigSurface()
let bigSurface = getBigSurface()
let da: DrawingArea = newDrawingArea()
da.setHexpand
da.setVexpand
da.connect("draw", daDrawing, bigSurface)
let
translateXAdj = newAdjustment(0, 0, 5000, 20, 0, 0)
translateYAdj = newAdjustment(0, 0, 5000, 20, 0, 0)
scaleAdj = newAdjustment(1, 1, 5, 0.1, 0, 0)
translateXLabel = newLabel("translate x")
translateXSpin= newSpinButton(translateXAdj, 50, 1)
connect(translateXSpin, "value-changed", translateXSpinChanged, da)
let translateYLabel = newLabel("translate y")
let translateYSpin = newSpinButton(translateYAdj, 50, 1)
connect(translateYSpin, "value-changed", translateYSpinChanged, da)
let scaleLabel = newLabel("Scale")
let scaleSpin = newSpinButton(scaleAdj, 0.2, 1)
connect(scaleSpin, "value-changed", scaleSpinChanged, da)
let grid = newGrid()
grid.attach(da, 0, 0, 3, 1)
grid.attach(translateXLabel, 0, 1, 1, 1)
grid.attach(translateYLabel, 1, 1, 1, 1)
grid.attach(scaleLabel, 2, 1, 1, 1)
grid.attach(translateXSpin, 0, 2, 1, 1)
grid.attach(translateYSpin, 1, 2, 1, 1)
grid.attach(scaleSpin, 2, 2, 1, 1)
add(window, grid)
showAll(window)
gtk.main()
main()A VTE example
The following code shows how we can use the vte library to create a plain shell window:
# https://vincent.bernat.im/en/blog/2017-write-own-terminal
import gintro/[gtk, glib, gobject, gio, vte]
proc appActivate(app: Application) =
let window = newApplicationWindow(app)
window.title = "GTK3 & Nim"
window.defaultSize = (600, 200)
let terminal = newTerminal()
let environ = getEnviron()
let command = environ.environGetenv("SHELL")
var pid = 0
echo terminal.spawnSync({}, nil, [command], [], {SpawnFlag.leaveDescriptorsOpen}, nil, nil, pid, nil)
window.add(terminal)
showAll(window)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", appActivate)
discard run(app)
main()A GStreamer example
Recently someone asked about gstreamer support for gintro, see https://github.com/StefanSalewski/gintro/issues/59 . So we added it. I don’t know much about gstreamer, but I was told that it is used with Python and lately with Rusts bindings, so there may be some use case.
# https://gstreamer.freedesktop.org/documentation/tutorials/basic/hello-world.html?gi-language=c
# nim c gstBasicTutorial1.nim
import gintro/gst
proc main =
var pipeline: gst.Element
var bus: gst.Bus
var msg: gst.Message
## Initialize GStreamer
gst.init()
## Build the pipeline
pipeline = gst.parseLaunch("playbin uri=https://www.freedesktop.org/software/gstreamer-sdk/data/media/sintel_trailer-480p.webm")
## Start playing
discard gst.setState(pipeline, gst.State.playing)
## Wait until error or EOS
bus = gst.getBus(pipeline)
msg = gst.timedPopFiltered(bus, gst.Clock_Time_None, {gst.MessageFlag.error, gst.MessageFlag.eos})
echo msg.getType
discard gst.setState(pipeline, gst.State.null) # is this necessary?
main()Threading Examples
A common problem with GTK GUI’s is the fact that functions that run for a longer time period in the GTK main thread can block the GUI for display updates or user input. We can solve this problem by creating a separate thread which runs these functions in the background.
The GTK GUI is not really thread safe, so we can not update the GUI from other threads directly. An easy way to solve this problem is to use the glib functions g-idle-add() or g-timeout-add().
The glib function g-timeout-add() adds a user defined function to the GTK main loop that is called periodically, and the function g-idle-add() adds a user defined function to the GTK main loop that is called when GTK is not busy with other stuff. Both functions are executed in the GTK main thread, so both can update GUI widgets or call other GTK related functions.
In our first example we create a Nim worker thread that sends data over a Nim channel to the main thread. We use timeoutAdd() to call periodically a function that reads data from the channel and updates the GUI. In this example the worker thread only does a plain countdown — in a real work example it would do the hard work in the background, i.e. running a chess engine to find the next best move or sorting a large data base. The function updateGUI() receives the current counter value and updates the label of a button.
# https://nim-lang.org/docs/channels.html
# nim c --threads:on --gc:arc -r thread1.nim
import gintro/[gtk, glib, gobject, gio]
from os import sleep
var channel: Channel[int]
var workThread: system.Thread[void]
proc workProc =
var countdown {.global.} = 25
while countdown > 0:
sleep(1000)
dec(countdown)
channel.send(countdown)
proc updateGUI(b: Button): bool =
let msg = channel.tryRecv()
if msg.dataAvailable:
b.label = $msg.msg
if msg.msg == 0:
workThread.joinThread
channel.close
return SOURCE_REMOVE
return SOURCE_CONTINUE
proc buttonClicked (b: Button) =
b.label = utf8Strreverse(b.label, -1)
proc activate (app: Application) =
let window = newApplicationWindow(app)
window.title = "Countdown"
window.defaultSize = (250, 50)
let button = newButton("Click Me")
window.add(button)
button.connect("clicked", buttonClicked)
window.showAll
channel.open
createThread(workThread, workProc)
discard timeoutAdd(1000 div 60, updateGUI, button)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", activate)
discard app.run
main()The next example uses the function g-idle-add() from inside the worker thread to call a user defined function that then updates a GUI widget:
# nim c --threads:on --gc:arc -r thread2.nim
import gintro/[gtk, glib, gobject, gio]
from os import sleep
var workThread: system.Thread[void]
var button: Button
proc idleFunc(i: int): bool =
button.label = $i
return SOURCE_REMOVE
proc workProc =
var countdown {.global.} = 25
while countdown > 0:
sleep(1000)
dec(countdown)
idleAdd(idleFunc, countdown)
proc buttonClicked(button: Button) =
button.label = utf8Strreverse(button.label, -1)
proc activate(app: Application) =
let window = newApplicationWindow(app)
window.title = "Countdown"
window.defaultSize = (250, 50)
button = newButton("Click Me")
window.add(button)
button.connect("clicked", buttonClicked)
window.showAll
createThread(workThread, workProc)
proc main =
let app = newApplication("org.gtk.example")
connect(app, "activate", activate)
discard app.run
main()A HeaderBar example for GTK4
The following code is the Nim version of a GTK4 example in C. It will work in its unmodified form only if you have GTK4 already installed — my GTK4 lives at /opt/gtk and I have to type these commands before I can use GTK4 programs like gtk4-demo:
export LD_LIBRARY_PATH=$LD_LIBRARY_PATH:/opt/gtk/lib64/ export GSETTINGS_SCHEMA_DIR=/opt/gtk/share/glib-2.0/schemas /opt/gtk/bin/gtk4-demo #export PKG_CONFIG_PATH="/opt/gtk/lib64/pkgconfig/" # only when compiling C code directly
The example is a bit complicated, as a custom (fake) headerbar is created, which involves a cast in C and Nim code. The example shows how to use a default headerbar, how to open a file dialog and how to use images and CSS styling. I guess most of the code would work in GTK3 also, but I have not tried to make a GTK3 version out of it, and I even do not understand all of the code yet. Note that for this example destroy() is not connected to the window close button, but called after gtk4.main(). A bit strange indeed, see forum note of Mr E.Bassi.
# https://github.com/GNOME/gtk/blob/mainline/tests/testheaderbar.c
# this is still based on the original testheaderbar.c, which was recently replaced by testheaderbar2.c
# and testheaderbar.c is not a very good Nim GTK4 example unfortunately -- too comlicated and strange code.
# nim c headerbar.nim
import gintro/[gtk4, glib, gobject]
const
Css = """
.main.background {
background-image: linear-gradient(to bottom, red, blue);
border-width: 0px;
}
.titlebar.backdrop {
background-image: none;
background-color: @bg_color;
border-radius: 10px 10px 0px 0px;
}
.titlebar {
background-image: linear-gradient(to bottom, white, @bg_color);
border-radius: 10px 10px 0px 0px;
}
"""
# we try to avoid use of global header variable as done in C code
type
MyWindow = ref object of gtk4.Window
header: gtk4.Widget
proc response(d: gtk4.FileChooserDialog; responseID: int) = gtk4.destroy(d)
proc onBookmarkClicked(button: Button; data: MyWindow) =
let window = gtk4.Window(data)
let chooser = newFileChooserDialog("File Chooser Test", window,
FileChooserAction.open)
discard chooser.addButton("_Close", gtk4.ResponseType.close.ord)
chooser.connect("response", response)
chooser.show
#proc changeSubtitle(button: Button; w: MyWindow) =
# if w.header.subtitle == "":
# w.header.setSubtitle("(subtle subtitle)")
# else:
# w.header.setSubtitle("") # can we pass nil?
proc toggleFullscreen(button: Button; window: MyWindow) =
var fullscreen {.global.}: bool
if fullscreen:
window.unfullscreen
fullscreen = false
else:
window.fullscreen
fullscreen = true
proc toIntVal(i: int): Value =
let gtype = typeFromName("gint")
discard init(result, gtype)
setInt(result, i)
var done = false
proc quit_cb(b: Button) = # we can not pass a var parameter
#gtk4.mainQuit()
done = true
wakeup(defaultMainContext()) # g_main_context_wakeup (NULL);
proc changeHeader(button: ToggleButton; window: MyWindow) =
if button != nil and button.getActive:
window.header = (newBox(gtk4.Orientation.horizontal,
10))
addCssClass(window.header, "titlebar")
addCssClass(window.header, "header-bar")
#window.header.setProperty("margin_start", toIntVal(10))
#window.header.setProperty("margin_end", toIntVal(10))
#window.header.setProperty("margin_top", toIntVal(10))
#window.header.setProperty("margin_bottom", toIntVal(10))
window.header.setMarginStart(10)
window.header.setMarginEnd(10)
window.header.setMarginTop(10)
window.header.setMarginBottom(10)
let label = newLabel("Label")
gtk4.Box(window.header).append(label)
let levelBar = newLevelBar()
levelBar.setValue(0.4)
levelBar.setHexpand
gtk4.Box(window.header).append(levelBar)
else:
window.header = newHeaderBar()
#addClass(getStyleContext(window.header), "titlebar")
addCssClass(window.header, "titlebar")
#window.header.setTitle("Example header")
var button = newButton("_Close")
button.setUseUnderline
addCssClass(button, "suggested-action")
button.connect("clicked", quit_cb)
gtk4.HeaderBar(window.header).packEnd(button)
button = newButton()
let image = newImageFromIconName("bookmark-new-symbolic")
button.connect("clicked", onBookmarkClicked, window)
button.setChild(image)
gtk4.HeaderBar(window.header).packStart(button)
window.setTitlebar(window.header)
proc main =
gtk4.init()
#var window: MyWindow
let window = newWindow(MyWindow)
addCssClass(window, "main") # gtk_widget_add_css_class (window, "main");
let provider = newCssProvider()
provider.loadFromData(Css)
addProviderForDisplay(getDisplay(window), provider, STYLE_PROVIDER_PRIORITY_USER)
changeHeader(nil, window)
let box = newBox(Orientation.vertical, 0)
window.setChild(box) # gtk_window_set_child (GTK_WINDOW (window), box);
#window.add(box)
let content = newImageFromIconName("start-here-symbolic")
content.setPixelSize(512)
content.setVexpand
box.append(content)
let footer = newActionBar()
footer.setCenterWidget(newCheckButtonWithLabel("Middle"))
let button = newToggleButtonWithLabel("Custom")
button.connect("clicked", changeHeader, window)
footer.packStart(button)
#var button1 = newButton("Subtitle")
#button1.connect("clicked", changeSubtitle, window)
#footer.packEnd(button1)
var button1 = newButton("Fullscreen")
footer.packEnd(button1)
button1.connect("clicked", toggleFullscreen, window)
box.append(footer)
window.show
#gtk4.main()
while not done:
discard iteration(defaultMainContext(), true) # g_main_context_iteration (NULL, TRUE);
destroy(window) # this is special for this example, see https://discourse.gnome.org/t/tests-testgaction-c/2232/6
main() # 137 lines| Related work: https://github.com/jdmansour/nim-smartgi |